Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Topical Pain Relief Using D-Amino Acid Hydrogels
Medicinal Chemistry: Hydrogel formulation of common drug could improve its selectivity and reduce its side effects
by Sarah Everts
November 28, 2012

For arthritis patients, nonsteroidal anti-inflammatory drugs can relieve pain. Unfortunately, the drugs also can cause serious side effects to the digestive tract, heart, and kidneys. In hopes of reducing side effects, chemists have now developed a supramolecular hydrogel made by coupling the common NSAID naproxen to short peptides based on D-amino acids (J. Am. Chem. Soc., DOI: 10.1021/ja310019x). This gel could lead to a topical formulation that patients would apply directly to painful joints.
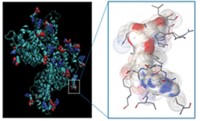
Supramolecular hydrogels are gels that form in water when small molecules self-assemble, says Bing Xu, a chemist at Brandeis University, who led the study. Xu’s group wondered if they could make a supramolecular hydrogel from a drug molecule, possibly producing a topical form of the drug.
The team formed hydrogels from naproxen, the pain drug sold under names such as Aleve, by adding short peptides consisting of aromatic D-amino acids to the drug’s carboxylic acid. Amino acids containing aromatic side groups, such as phenylalanine, are known to promote hydrogel formation, Xu says. His group chose D-amino acids because the molecules cannot be broken down by the body’s enzymes that typically target L-amino acids. Thus, the researchers thought, the hydrogel would last long enough to allow naproxen to relieve pain by inhibiting the protein cyclooxygenase-2 (COX-2).
In solution-based tests, the chemists determined how well each drug-peptide complex inhibited COX-2 and cyclooxygenase-1 (COX-1), the protein involved in many of naproxen’s side effects. The hydrogels were 20 times more selective at shutting down COX-2 than at inhibiting COX-1, Xu says.
The team hadn’t expected to see improved selectivity, Xu says, but he thinks it should help limit side effects.
Garret A. FitzGerald, who studies COX enzymes at the University of Pennsylvania, calls the new hydrogels “a nice bit of chemistry,” particularly because they reduce inhibition of COX-1 compared to COX-2. But he adds that the researchers have a long way to go before they’ll know whether the gels could work in the clinic.
The Brandeis team next wants to test whether coupling D-amino acids to other drugs also reduces their side effects.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter