Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Nanocoating Helps Furniture Foam Fight Fire Without Troubling Flame Retardants
Nanomaterials: An ultrathin film made from a biopolymer and a sulfur-based acid could be an effective replacement for chlorine- and bromine-based commercial flame retardants
by Prachi Patel
April 30, 2013

A 30-nm-thick polymer coating can prevent the spread of flames on foam used in home furnishings (ACS Macro Lett., DOI: 10.1021/mz400105e). The coating, which consists of a sulfur-based polymer and a carbohydrate polymer found in crustacean shells, could be an environmentally friendly alternative to the flame retardants used today in bedding and sofas, say its developers.
Mattresses and furniture cushions are typically made of highly flammable polyurethane foam. To meet fire safety guidelines, manufacturers treat the foam with fire-retardant chemicals, which are typically brominated compounds. Toxicologists and environmental scientists worry about these chemicals because previous studies have shown that some can act as endocrine disruptors, lead to neurological problems, or even cause cancer. The European Union has banned several of the flame retardant compounds, and regulatory agencies in the U.S. and Canada have started to scrutinize the chemicals’ use.
The compounds also affect the foam’s stiffness. Adding more of the chemicals to foam increases the material’s fire resistance but makes upholstered furniture less comfortable, says Jaime C. Grunlan, a mechanical engineering professor at Texas A&M University.
Grunlan wants to find flame-retardant compounds that do not affect the foam’s properties and are less toxic than current formulations.
He and his colleagues have developed a new coating made from polyvinylsulfonic acid (PVS) and chitosan, a long carbohydrate molecule derived from the shells of shrimp and other crustaceans. Neither polymer contains a potentially harmful brominated compound.
To produce the coating, the researchers dip foam alternately in water solutions of the two polymers. Chitosan is positively charged in water, while PVS is negatively charged. The two oppositely charged polymers attract each other and get entangled, creating the coating.
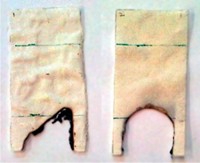
The 30-nm-thick film increases the weight of the foam by just 5.5% and doesn’t alter the foam’s stiffness. The researchers tested the flame retardant properties of the coating by exposing treated foam to a flame from a butane torch for 10 seconds. While uncoated foam burned up completely, the fire on the coated foam went out once the researchers turned off the torch. It would take more than three times as much conventional flame-retardant material by weight to achieve the same effect, Grunlan says.
He explains that when the PVS burns it gives off vapors of sulfur oxides, which are nonflammable. “This creates a gas blanket on the foam surface, cutting off oxygen and starving the fire,” he says. The researchers had assumed that the chitosan would release nitrogen and water, creating a char on the foam surface that would act as a heat shield between the flame and the foam. But they don’t see evidence of that.
Still Grunlan says the nanocoating needs chitosan as a positively charged polymer to combine with the negatively charged PVS. He adds that there might be alternatives to chitosan that could enhance the coating’s flame retardant properties.
The nanocoating could be sprayed on foam, which would make it easy for mass production, Grunlan says. Several companies have expressed interest in the material, he adds.
Rick D. Davis, leader of the flammability reduction group at the National Institute of Standards and Technology, says fireproof nanocoatings are cutting-edge technologies in the field. He works on clay-based ones. Grunlan’s nanocoating is unique because it’s the first to use sulfur-based gas to extinguish fire, Davis adds. He says that nanocoatings, both his and Grunlan’s, are more effective and could be less toxic than commercially available flame retardants.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter