Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Small-Ring Substitute Helps Drugs Stick Around
Medicinal Chemistry: Trifluoromethylcyclopropyl groups could replace tert-butyl groups on potential medications that don’t last in the bloodstream
by Carmen Drahl
May 1, 2013

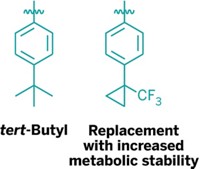
The human body’s metabolism has a way of holding up the search for a new drug. Metabolic enzymes react with certain groups on a molecule, modifying the compound and preventing it from lasting long enough in the body to be useful. One vulnerable target is the tert-butyl group. A new replacement for the group could extend potential medications’ staying power (ACS Med. Chem. Lett., DOI: 10.1021/ml400045j).
tert-Butyls aren’t always trouble. “It really depends on the context,” says medicinal chemist David Barnes-Seeman. Some drugs on the market feature a tert-butyl group, including the enlarged prostate drug finasteride, and are just fine. However, tert-butyls are vulnerable to cytochrome P450 enzymes in the liver. These enzymes oxidize methyl groups in the tert-butyl, hastening a molecule’s excretion from the body.
Barnes-Seeman’s group at the Novartis Institutes for Biomedical Research, in Cambridge, Mass., ran into this very problem with a drug lead they study. They tried to tweak the tert-butyl to make it more polar, an established approach for helping a molecule stay around longer in the body. Some of these changes didn’t work, and those that did reduced the compounds’ binding affinity for their target protein, an unacceptable compromise.
They then turned to trifluoromethylcyclopropyl groups, which resemble a tert-butyl but lack vulnerable methyl groups. Other researchers have tried this replacement to alter a molecule’s binding affinity for a protein, Barnes-Seeman says, but they hadn’t reported its metabolic stability.
To test the tert-butyl analog, his group measured the lifetime of drug compounds in the bloodstreams of rats. Compounds with the tert-butyl replacement held out longer than those containing a tert-butyl—about four times longer for one compound. In addition, the compounds did not lose their binding affinity for their protein target.
This tactic is “a reasonable alternative” for dealing with tert-butyl groups, says cytochrome P450 expert F. Peter Guengerich at Vanderbilt University. The new functional group may not always be so innocuous to binding affinity, he cautions, “so I don’t know that I’d start sticking this everywhere.”
This strategy isn’t a panacea, agrees Barnes-Seeman. But he thinks chemists will want to add it to their toolkits anyway. “If you have a tert-butyl group you’re struggling with,” he says, “just try switching it out for cyclopropyl-CF3 and see if it works for you.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter