Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
New Compound Stops Flu Virus’s Thievery
Medicinal Chemistry: Molecule prevents virus replication by inhibiting a viral enzyme that steals host RNA
by Olga Kuchment
September 10, 2013

In hopes of adding new weapons to the antiflu arsenal, researchers have set their sights on an enzyme that helps the virus replicate in host cells by stealing RNA. The team now reports a new class of molecules that inhibit this enzyme and prevent influenza from replicating in mammalian cells (ACS Chem. Biol. 2013, DOI: 10.1021/cb400400j).
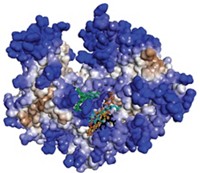
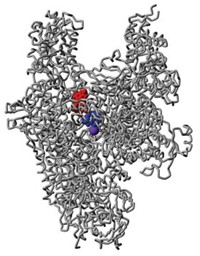
The enzyme, called polymerase acidic protein (PA), is a part of the virus’ polymerase complex. It clips off a short portion at the beginning of host messenger RNAs floating around the host’s nucleus. The polymerase then uses these short sequences to prime transcription of the virus’ genes. A portion of the enzyme, called the cap-snatching endonuclease domain, uses two metal ions to perform this clipping. Joseph D. Bauman, Kalyan Das, and Eddy Arnold of Rutgers University had been designing inhibitors for metal-binding enzymes in viruses such as HIV, and realized that the PA endonuclease would be a prime target for an antiviral drug.
Arnold’s team first crystallized the endonuclease domain from the 2009 H1N1 virus in a way that allowed inhibitors to seep into the active site, allowing the researchers to analyze how well the molecules fit. After screening hundreds of small molecules, the most promising one was a hydroxypyridinone compound. The team worked with collaborators to synthesize a series of derivatives and test their abilities to block the enzyme’s activity.
The best inhibitor slowed viral replication in cultured dog kidney cells by 50% at a concentration of 11 µM. Crystal structures of the enzyme-inhibitor complex showed that the molecule not only binds the two metal ions in the active site, but also a third one that could be essential for enzyme activity.
The team hopes to test this inhibitor and new ones in animals. Developing new antiflu compounds could help people stay ahead of the evolving virus, Arnold says. “Having only one good oral drug for influenza, Tamiflu, is not enough,” he says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter