Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Delivery
Eyes On Excipients
Inactive but essential drug ingredients are getting checked out with modern rigor to ensure their quality
by Jyllian Kemsley
February 3, 2014
| A version of this story appeared in
Volume 92, Issue 5

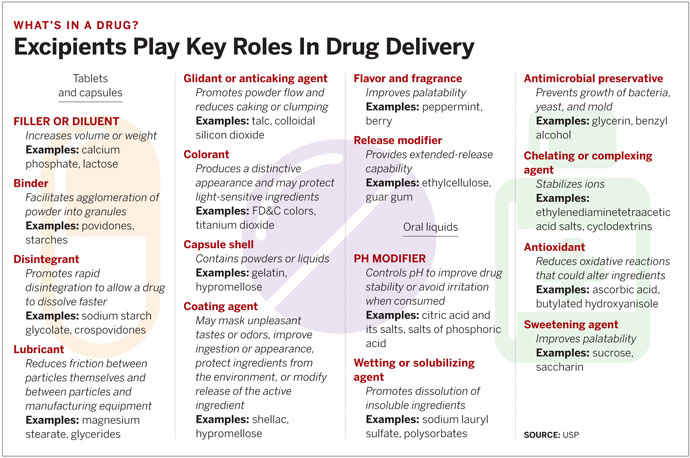
When it comes to drugs, most of the attention goes to the active pharmaceutical ingredient. But typically the vast majority of what’s in a tablet, capsule, or liquid isn’t the active ingredient. A drug is formulated with many other compounds, called excipients, that play an essential role in delivering the active ingredient to the patient.
Lubricants, for example, make a bulk material easier to handle in a manufacturing facility. Fillers may be added to make small doses, such as 5 mg of an antihistamine, physically large enough for a person to handle easily. Fillers may double as binders to help hold a tablet together. Coatings protect a tablet from moisture or light or mask an unpleasant flavor. Disintegrants help a tablet fall apart in the digestive tract to release the active drug.
The pharmaceutical-standards-setting organization U.S. Pharmacopeia (USP) counts nearly 500 excipients in its National Formulary. The global excipients market was $5.9 billion in 2012 and is expected to reach $8.8 billion in 2018, according to market research firm BCC Research.
Despite the crucial role that excipients play in getting drugs successfully into patients, some regulatory analytical procedures for excipient quality control are decades old. These methods do not meet modern expectations for analytical rigor or actually test that a product is what’s said on its label. What’s more, excipients are not exempt from rising anxiety in recent years about the integrity of global pharmaceutical supply chains. In one instance, counterfeiters substituted less expensive and toxic diethylene glycol for glycerin in cold medicine sold in Panama in 2006, killing at least 100 people. In response to these concerns, USP, in collaboration with the U.S. Food & Drug Administration and similar organizations around the world, is working on updating analytical requirements for excipients.
It’s an effort that’s easier said than done. Many excipients are not cleanly synthesized small molecules. Instead, they may be analytically challenging materials that are polymeric, prepared from animal or plant material, or mined. Also, the markets for industrial or food uses for a particular product often far outweigh the pharmaceutical excipients market. Manufacturers may not see a strong need to continue to supply the pharmaceutical industry if they’re faced with buying expensive instrumentation that requires highly skilled operators. Raising analytical standards is consequently a balancing act between ensuring the safety of pharmaceuticals and preserving the excipient supplies to formulate them.
“More robust acceptance criteria serve as a competitive advantage for high-quality excipient producers,” because the standards assure pharmaceutical companies that they will be able to make finished products free of contaminants or adulterants, says Carl Perini, manager of quality compliance for Ashland Specialty Ingredients.
At the same time, “if we raise the bar too high, people will drop out,” says R. Christian Moreton, founder of FinnBrit Consulting, which focuses on drug development. That’s a particular concern for excipients that are not widely used. Loss of an excipient from the market would mean a time-consuming and expensive effort to reformulate a drug and get FDA approval for it.
One way to approach the problem of modernizing U.S. standards is to take inspiration from the techniques other countries require and manufacturers may already be using, says Catherine M. Sheehan, USP’s director of excipient standards. For example, USP recently updated the testing of guar gum, which is a polysaccharide milled from guar beans that is used in foods as a thickening agent. In pharmaceuticals, guar gum serves as a disintegrant and release-modifying agent for extended-release tablets. FDA identified guar gum to be at risk for adulteration with products such as less costly locust bean gum because of lack of testing specificity.
USP eventually decided to adopt a procedure used by the European Pharmacopeia, in which the gum is hydrolyzed and the major mannose and galactose components are identified by thin-layer chromatography. Some guar gum manufacturers were not happy about having to implement the new technique, Sheehan says, “but we’re not talking about using high-performance liquid chromatography or nuclear magnetic resonance spectroscopy. The method requires minimal cost and minimal expertise.”
Another excipient in need of updating was gelatin. Gelatin’s major industrial use is for glue, but it also serves as a gelling or thickening agent in foods. Pharmaceutical companies use it to make capsules. Gelatin is hydrolyzed collagen; it is prepared by treating animal hides or bones with acid or base, then extracting the collagen with water or acid.
Traditionally, the main quality-control attribute for gelatin is something known as “Bloom strength,” named for developer O. T. Bloom. The test involves forming a 6.67% gelatin gel, then seeing how much force is necessary to depress it by 4 mm. “Is it a great scientific test? No, but it works, and industry has adjusted to it,” Moreton says.
But the test doesn’t say whether the material is, in fact, gelatin and only gelatin. So USP decided to test not only for gelling but also for protein using a common assay that yields a color change when protein binds to copper sulfate. “If it’s protein and it gels, then it’s likely gelatin because not many proteins gel in that way,” Moreton says.
In a Dec. 13, 2013, letter to USP, however, FDA expressed concern that gelatin might be subject to adulteration with melamine, a substance that causes kidney failure and was used to inflate apparent protein content in milk products in China in 2008. Although studies by the Gelatin Manufacturers Institute of America indicate that adding melamine to gelatin impairs gelling, FDA still wants gelatin tested for melamine. The agency suggested powder X-ray diffraction, near-infrared spectroscopy, and Raman spectroscopy as possible approaches.
USP is mulling how to respond. X-ray diffraction is neither inexpensive nor quick, both problems in a quality-control setting, comments Moreton, and neither infrared nor Raman spectroscopy is as reliable as analysts would like. “You’d have to make sure you can see absence of melamine and that there’s no interference from any of the gelatin peaks in different types of gelatin,” he says. USP plans to discuss the matter with stakeholders at a meeting in June.
Melamine adulteration is also a concern for povidones, a family of polyvinylpyrrolidone polymers with varying molecular weights. Related materials include copovidones, which are copolymers composed of N-vinylpyrrolidone and vinyl acetate, and crospovidones, which are cross-linked polyvinylpyrrolidones. Nonpharmaceutical uses of these polymers include hair styling, skin care, membranes for medical devices and water treatment, and adhesives. Crospovidone is used to absorb polyphenols to clarify beer. In pharmaceuticals, both povidones and copovidones function as binders. Copovidones are also used in coatings and to provide a soluble matrix for melt-extruded formulations. Crospovidones serve as disintegrants.
Current analytical quality-control tests for povidones and related polymers involve IR spectroscopy and reactions to look for colored complexes formed by the ends of the polymers. Additionally, analysts look for nitrogen using the Kjeldahl test, which involves digestion of a sample in sulfuric acid. Unfortunately, melamine and other materials of concern for adulteration of povidones, such as urea and nylon 6,6, also contain nitrogen, Ashland’s Perini says. Consequently, the Kjeldahl test won’t sniff them out.
As a USP expert panel looks to modernize analysis of the polymers, a particular challenge is that crospovidone is insoluble. One promising test the group has looked at is to evaluate the mass fractions of carbon, hydrogen, nitrogen, and any heteroatoms after combustion of a sample, says Bernhard Fussnegger, who works in global marketing for pharma and medical ingredients at BASF. The ratios enable determination of the elementary composition of the material, independent of the solubility of the polymer matrix and potential adulterants. The group is also considering tests such as pyrolysis-gas chromatography and high-performance liquid chromatography.
Another excipient under USP’s modernization microscope is talc, which is used in pharmaceuticals as a filler, anticaking agent, lubricant, and coating agent. Other applications include plastics, paints, and ceramics, as well as personal care products and food. Talc is a hydrous magnesium silicate mineral, Mg3Si3O10(OH)2, that forms into sheets held together by weak van der Waals interactions. The layered plates are what give talc its unique soft and slippery feel, says Julie Pier, analytical laboratory manager for filtration and performance additives at mineral product manufacturer Imerys.
Talc is mined and can form from several different types of source rock, each with its own signature elemental and mineralogical composition. Depending on the host rock type, impurities, and factors such as temperature and pressure, talc can co-occur with asbestos. Asbestos minerals form into characteristic small fibers and are best known for causing lung disease, although asbestos is also associated with gastrointestinal cancers. U.S. talc supplies historically have been asbestos-free, but asbestos-contaminated talc has turned up in China and South Korea.

Elemental analysis cannot be used to identify asbestos, Pier says, because asbestos and nonasbestos crystal morphology can form from the same minerals. The existing USP method for detecting asbestos in talc relies largely on X-ray diffraction (XRD) to determine whether a possible asbestos mineral is present in a sample. If one is picked up by XRD, then polarized light microscopy is used to check for asbestiform morphology.
But the limit of detection for XRD is about 0.5%. Analysts would like to get to 0.1% or lower. The USP panel working on the problem thinks that XRD is still a good screening test to give a general sense of the overall purity of a sample, says Pier, who spoke about the panel’s work at USP’s Science & Standards Symposium held in Baltimore in September 2013.
To get around the limit of detection problem, the panel may require that XRD be coupled with a microscopy method: polarized light, scanning electron, or transmission electron.
“With polarized light microscopy you can see more material, so you have a better chance of detecting a fiber, but the trade-off is lower resolution,” Pier says. In contrast, transmission electron microscopy has very high resolution, but it looks at such a small field of material that finding fibers is statistically difficult. Scanning electron microscopy falls somewhere in between. The techniques also have other strengths and weaknesses. “Not one method is clearly the best, regardless of cost,” Pier says. “Even if you have $1 million to spend, you wouldn’t necessarily choose one method over another.”
The answer may be to apply a sieve method used at Imerys that takes advantage of physical properties of the minerals: Talc platelets are easy to grind into small particles, whereas asbestos fibers are not. Filtering ground material through an inexpensive sieve allows for detection of asbestos at less than 0.1%, depending on the type of source mineral, because the hard-to-grind fibers are retained, Pier says.
The USP talc panel hopes to publish a summary of its work later this year to stimulate discussion in the community, then work on developing specific procedures.
Overall, USP modernized its testing monographs for 51 excipients in the years 2005–10. It hopes to complete 75 more by 2015. USP normally relies heavily on stakeholders to serve on its expert panels, suggest analytical approaches, and provide samples for procedure development and validation. “For high-volume excipients that are used in a lot of drug products, there is tremendous support from stakeholders on modernizing,” USP’s Sheehan says. Engaging excipients manufacturers of lower-volume materials has been much more challenging. That has led to an expansion of USP’s own labs, as the organization fills the gap.
Ultimately, all the work to modernize excipients analysis will benefit patients in the form of a safer drug supply. Well-characterized excipients with consistent quality allow pharmaceuticals to be manufactured with reliable efficiency and stability, BASF’s Fussnegger says, adding, “Without excipients, the pharmaceutical industry would not be where it is today.”






Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter