Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Engineered Bacteria Stick To Cancer Cells
Synthetic Biology: Researchers modified bacterial proteins called adhesins to target proteins expressed on human cells
by Erika Gebel Berg
August 18, 2014

It’s usually bad news when a swarm of foreign bacteria pursues cells in the body. But some scientists want to deploy engineered microbes to specific parts of the body to kill tumor cells or deliver drugs. Researchers have now designed a way to target bacteria to particular surfaces and tissues by modifying sticky proteins on the microbes’ surfaces (ACS Synth. Biol. 2014, DOI: 10.1021/sb500252a).
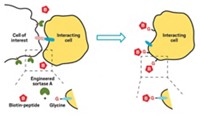
Scientists can develop antibodies to target virtually any antigen. So a few years ago, Luis Ángel Fernández of the Spanish National Center for Biotechnology and his group explored the idea of making bacteria seek out certain other cells by engineering the microbes to display antibodies on their surface. Unfortunately, getting the cells to deliver antibodies to their surfaces proved difficult. Fernández then learned about adhesins, proteins that naturally coat the surface of bacteria. The adhesins contain an antibody-like moiety that targets antigens on other cells, facilitating biofilm formation, cell invasion, and other pathogenic activities. Fernández realized that he could potentially swap out the natural antibody-like domain for another that is specific to a molecule of choice, allowing him to target bacteria to particular biological surfaces.
In a series of proof-of-principle experiments, Fernández’s team tested whether they could make Escherichia coli target human cancer cells. The researchers built a synthetic adhesin with an antibody that binds to green fluorescent protein (GFP). They incorporated the new adhesin’s gene into the chromosome of E. coli, along with a gene for a bioluminescent protein to help detect the bacteria. The team also deleted the genes for three natural adhesins to reduce nonspecific binding.

The bacteria’s targets were human cancer cells that the researchers modified to express GFP on their surfaces. After mixing the human and bacterial cells together, the team inspected the cells with fluorescence imaging and found that the bacteria colonized the cancer cells. Bacteria without the synthetic adhesin did not adhere to the human cells.
To test their approach in live animals, the researchers used the same GFP-bearing human cancer cells to grow tumors in the abdomens of nine hairless mice. Then, they injected approximately 100,000 bacteria with the synthetic adhesin into the tails of the mice. Four days later, eight of the nine tumors glowed from the bioluminescence signals of the bacteria. They repeated the experiment using bacteria that expressed the bioluminescent protein but not the adhesin. Only two out of nine tumors were colonized.
This approach “gives rise to an array of applications,” says Matthew W. Chang of the National University of Singapore. However, the researchers still need to demonstrate safety and that the bacteria can be engineered to target real-world cancer cell proteins. Fernández is currently developing synthetic adhesins that target proteins in bladder and gastrointestinal cancers. These diseases represent what may be the safest application of this technology, he says, because they occur in parts of the body normally inhabited by bacteria.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter