Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Liquid Crystals Stabilize Dye-Sensitized Solar Cells
Photovoltaics: New electrolyte lets dye-sensitized solar cells work at high temperatures
by Neil Savage
November 7, 2014

Dye-sensitized solar cells (DSCs) could lead to inexpensive, easy-to-manufacture photovoltaics. But they have one main drawback: They don’t work very well at high temperatures, which can be a problem for a device designed to absorb sunlight. Now a group of researchers has incorporated liquid crystals into the devices, producing a version that works at up to 120 °C, the highest reported working temperature for such solar cells (Chem. Mater. 2014, DOI: 10.1021/cm503090z).
DSCs, also known as Grätzel cells, consist of an anode and a cathode separated by an electrolyte. The anode carries particles of titanium dioxide, coated with a dye, that absorb sunlight and then eject electrons. Iodide ions in the electrolyte help transport electrons from the cathode back to the anode, creating an electric potential.
In early DSCs, the electrolyte consisted of an organic solvent containing iodide ions. Unfortunately, the liquid tended to leak and evaporate at high temperatures. More recent DSCs use nonvolatile electrolytes such as polymers, but these devices are not efficient at converting sunlight into power. Ionic liquids, which have high conductivities and low volatility, make promising electrolytes but become unstable at high temperatures.
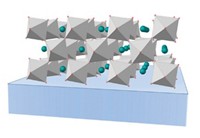
So Takashi Kato of the University of Tokyo and his colleagues decided to create a more stable electrolyte by combining an iodide-based ionic liquid with a carbonate-based molecule that forms liquid crystals. The liquid crystals self-assemble into a layered nanostructure, allowing the researchers to design channels in the electrolyte that transport ions efficiently. Unlike the liquid crystals used in displays, these are not designed to change their orientation in response to a voltage. Because the liquid crystals remain stable at high temperatures, the combination electrolyte stays viable as well.
The solar cells converted 2.7% of collected light into electricity at 90°C. Standard dye-sensitized solar cells are 11 to 12% efficient at room temperature, but their performance falls off above 50 °C. International standards call for solar cells to be tested at 85 °C. Kato hopes to increase the efficiency of his cells to 8 to 10%, which should be enough to make them commercially viable.
He plans to experiment with different combinations of materials in the electrolyte to try to increase performance. It should also be possible, he says, to polymerize the electrolyte after the liquid crystals organize, locking an optimal nanostructure into place. Such a solid-state electrolyte would never have a problem with leakage, he explains, and would allow for more flexible solar cells.
Erik Spoerke of Sandia National Laboratories says the devices probably wouldn’t regularly run at 120 °C, but “higher temperature stability means the devices would likely be less susceptible to degradation at more reasonable operating temperatures.”
Edwin C. Constable, a chemist at the University of Basel, in Switzerland, says the new electrolyte “opens up new ways to further improve the performance of DSCs.” Catherine E. Housecroft, also of Basel, says that using “liquid crystals to provide a matrix for ion transport is indeed an exciting development and one that should be of wide interest to the DSC community.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter