Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
New Approach To Control Enzyme Function In Cells
Molecular Biology: Technique switches enzyme activity on and off with light
by Stu Borman
May 22, 2015

A new technique could make it easier to target and inhibit specific enzymes and other proteins in cells and to turn that regulation on and off at will with light. The approach provides researchers with another tool to study the function of proteins in their natural cellular environments.
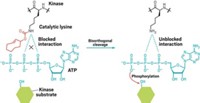
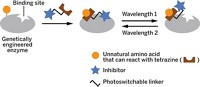
In the study, Jason W. Chin at the Medical Research Council Laboratory of Molecular Biology, in Cambridge, England, and coworkers, selectively inhibit enzymes by covalently tethering small-molecule inhibitors to target proteins. They do this with the help of a technique they previously reported that involved genetically encoding proteins in cells with unnatural amino acids bearing strained alkene and alkyne groups. The researchers could then rapidly and specifically label the proteins with tetrazine derivatives that react with the strained chemical groups.
Chin’s team has now used this labeling strategy to develop bioorthogonal ligand tethering, or BOLT, and has demonstrated its use to create an inhibitor that can regulate the activity of a target enzyme selectively, an approach they call iBOLT (Nat. Chem. 2015, DOI: 10.1038/nchem.2253).
To use iBOLT, the researchers engineer a strained alkyne into an enzyme in cells and then add a tetrazine-inhibitor conjugate to the cells. An alkyne-tetrazine reaction then tethers the inhibitor in close proximity to its binding site on the target protein, leading to selective or specific inhibition.
They demonstrated iBOLT by using it to specifically inhibit each of two closely related signaling enzymes, the kinases MEK1 and MEK2, in live mammalian cells. Problems occur in the enzymes’ signaling pathway in 30% of human tumors.
The paper also reports photo-BOLT, which involves using a photoswitchable linker to synthesize the tetrazine-inhibitor conjugate. This connector changes shape with different wavelengths of light, allowing the researchers to turn inhibitor binding, and consequently enzyme inhibition, on and off at will.
BOLT’s limiting factor is the need for engineered expression of unnatural amino acids, comments Anthony C. Bishop of Amherst College, an expert on target-specific control. Other methods for inhibiting kinase enzymes selectively require less extensive cell engineering, he says. However, “there are not, to my knowledge, other methods for reversibly controlling their activities with the spatiotemporal control that light affords,” he says. “The authors’ data that demonstrate the reversible control of MEK1 activity by simply switching the wavelength of light are quite beautiful.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter