Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Dual C–F/C–H functionalization unveiled
Iridium photocatalyst enables coupling of two unactivated aryl partners to create complex multifluorinated biaryl compounds
by Stephen K. Ritter
March 7, 2016
| A version of this story appeared in
Volume 94, Issue 10
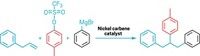
In an effort to develop a general method for making multifluorinated biaryl compounds, a pair of chemists has achieved the first example of a direct cross-coupling reaction between a C–F bond of one aryl compound and a C–H bond of another aryl compound without having to first prefunctionalize either of the aryl coupling partners like in traditional cross-coupling strategies (J. Am. Chem. Soc. 2016, DOI: 10.1021/jacs.5b13450). Sameera Senaweera and Jimmie D. Weaver of Oklahoma State University, Stillwater, previously determined how to use an iridium photocatalyst with amine reductant to selectively functionalize a C–F bond of multifluorinated aryl compounds. Along the way they realized there was an opportunity to go further with the perfluoroaryl radical intermediate and use it to couple with a nonfluorinated aryl partner if they could create the right reaction conditions. Starting from pentafluoropyridine, they found that the iridium catalyst with a bulky amine under blue LED light works, leading to a wide array of multifluorinated biaryl compounds (one example shown) that are typically useful in drugs, agrochemicals, and electronic materials. “We believe that, even though some of the yields are modest, the fact the reaction takes place in a single step from two broadly available partners and tolerates a broad range of functionality really gives this method legs,” Weaver says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter