Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Chemists engineer metalloproteins with novel activities
New protein engineering method yields proteins capable of catalyzing reactions not seen before in biology
by Stu Borman
June 16, 2016
| A version of this story appeared in
Volume 94, Issue 25
A new technique allows researchers to create artificial metalloproteins that can catalyze reactions distinct from those of any natural protein.

The technique could “expand the toolbox of biocatalytic strategies available to chemists for asymmetric synthesis of high-value compounds such as fine chemicals and pharmaceuticals,” says Rudi Fasan of the University of Rochester, who was not involved in the work.
Chemists have previously reported methods for producing new metalloproteins, and the new strategy combines key advantages of two of those techniques.
Frances H. Arnold of Caltech and others pioneered the use of directed evolution, an iterative protein modification technique, to revise key amino acids in a metalloprotein to tweak its chemistry. But this method does not include changing the protein’s metal center to attain novel types of activity.
Another method allows for such metal center changes. It starts with a protein that lacks a metal center and then creates one by inserting a metal-cofactor complex into the binding or active site. But introducing these metal centers changes the protein’s ability to bind substrates in unexpected ways and thus makes it difficult for researchers to identify key binding-site amino acids to modify. Directed evolution has therefore not worked well with this method.
John F. Hartwig and coworkers at the University of California, Berkeley, have now melded these two techniques by replacing a metalloprotein’s native metal with a nonnative one (Nature 2016, DOI: 10.1038/nature17968). This perturbs the binding site much less than when a new metal is inserted into a non-metalloprotein. As a result, the team can more readily use directed evolution methods to further change the protein’s chemistry. Scientists have replaced metals in metalloproteins before, but in those cases the teams generally didn’t seek or obtain significantly new forms of reactivity.
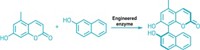
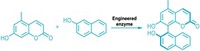
In the new study, Hartwig’s group expressed in bacteria a form of myoglobin lacking its iron-heme unit. They reconstituted it with various metal porphyrins and found iridium to be the most active metal center for the type of reactivity they were seeking. They then used directed evolution to produce many versions of the iridium-substituted metalloprotein.
Some of the iridium proteins catalyze reactions that add carbenes to alkenes and α-olefins, and others convert C–H bonds into C–C bonds by enantioselective carbene insertion—the first time any proteins have catalyzed these types of reactions.
Arnold says the new work offers the exciting ability to combine protein engineering and directed evolution to create proteins with new chemistries and the exquisite selectivity enzymes offer. “A world of possibilities for nonnatural chemistry is now open,” she says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter