Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biomaterials
Sponge made of coffee grounds scrubs lead and mercury from water
The material could make it easier to use coffee waste for water remediation
by Prachi Patel
September 21, 2016

Homes, restaurants, and the coffee industry collectively produce about 6 million tons of spent coffee grounds every year. Researchers have now come up with a practical way to use some of this waste. They have made a rubbery foam from used coffee powder and silicone that can pull lead and mercury ions from water (ACS Sustainable Chem. Eng. 2016, DOI: 10.1021/acssuschemeng.6b01098). The spongelike material could be used to clean heavy-metal-contaminated water on a large scale, its creators say.
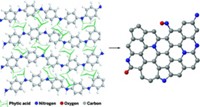
Spent coffee grounds are already used as fertilizer and converted into biodiesel. Scientists have also studied the ability of coffee grounds to remove heavy metals from wastewater, since the grounds contain charged amine, carboxylic, and phenolic groups that adsorb heavy metal ions. But filtering grounds out of the water postcleanup is difficult.
So Despina Fragouli and her colleagues at the Italian Institute of Technology decided to fix coffee into a rubbery, porous material—a silicone foam that is cheap and easy to make.
The researchers made the foam by mixing finely ground used coffee powder and a small amount of sugar into a solution of the elastomer acetoxy polysiloxane and a polydimethylsiloxane surfactant, and allowing the mixture to dry and polymerize overnight. Then the researchers dipped the material into warm water to dissolve the sugar crystals, leaving behind pores and yielding a spongy foam that contains 60 to 70% coffee by weight.
To test its ability to clean wastewater, the team immersed the foam in aqueous solutions of varying concentrations of lead and mercury ions for 30 hours. Each gram of the foam could adsorb a maximum of about 13 mg of lead ions and up to 17 mg of mercury. The foam adsorbed more than five times as much lead by weight as spent coffee powder. “This makes it realistic to use spent coffee in a large scale application,” Fragouli says. By using a sufficient amount of foam, it should be possible to remove enough metal ions to meet drinking water standards, she adds.
“We are exploring ways to remove metal ions from the foams without altering their functionality so they can be reused,” Fragouli says. The researchers are also planning to make fully biodegradable foams to make disposal simpler and more cost effective. The team has also begun making the foam from elastomers that spontaneously become porous during polymerization by forming gas bubbles, eliminating the need for sugar in the process.
No one has attempted to make composite coffee foams for water remediation before, says Constantine M. Megaridis, a mechanical engineer at the University of Illinois, Chicago. “Millions of tons of spent coffee wind up in landfills every year, so the proposed method not only reduces the solid waste stream but removes dangerous heavy metal pollutants from water,” he says. But collecting enough coffee grounds for large-scale application of this technology could be difficult, he adds, and doing it cost effectively would require a system to be set up with commercial users like hotels and restaurants.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter