Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Potential diabetes treatment uses light-activated gel to release insulin
Injectable insulin-carrying polymer could offer a less invasive way to control blood sugar
by Jyoti Madhusoodanan
October 27, 2016

Patients with type 1 diabetes rely on daily injections of insulin to help regulate their blood glucose in response to food, exercise, or other activities. Now, researchers have devised a potentially less invasive way to deliver the drug: an insulin-carrying gel that can be placed under the skin and activated by a flash of light to release the hormone when needed (Mol. Pharmaceutics 2016, DOI: 10.1021/acs.molpharmaceut.6b00633).
Because the body’s insulin needs vary over the course of a day, and from one day to the next, finding the right dose can prove difficult. When blood glucose rises after a meal, the pancreas releases insulin so cells can absorb and utilize the sugar. When glucose dips, insulin secretion slows. Efforts to treat type 1 diabetes aim to mimic the pancreas’s sensitive response to blood glucose levels. Current FDA-approved artificial pancreas devices consist of a continuous glucose monitoring system and an external insulin pump, which releases small amounts of the hormone into the blood as needed. But external pumps deliver insulin through tubes inserted under the skin, which pose infection risks and need to be replaced regularly. The new method, which relies on light shone on the skin to activate insulin release, avoids these drawbacks. “We’re trying to design an alternative to the pump,” says study author Simon H. Friedman of the University of Missouri, Kansas City.
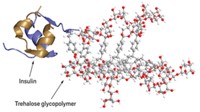
To make the material, Friedman and his colleagues linked human insulin molecules to a commercially available gel—a polystyrene and polyethylene glycol copolymer—using a linker molecule containing a light-sensitive chemical bond. When exposed to 365-nm-wavelength light, this bond breaks, releasing the insulin.
To test the material, the researchers injected a total of 80 μL of the gel, in the form of 10-μm-diameter beads, under the skin of diabetic rats and attached a coin-sized light-emitting device over the area. The team switched on the light for two minutes and then measured the animals’ insulin and blood glucose levels every 15 minutes, using an enzyme assay for the insulin and a standard glucometer. Insulin release began five minutes after the light exposure, peaked at approximately 25 minutes, and then plateaued. In control animals—which had light blocked from the skin by a layer of aluminum foil—the polymer did not release insulin. Blood glucose levels dropped in experimental animals in response to the hormone, but remained high in the control group. The researchers reactivated the gel 65 minutes later to release a second dose of insulin. “This is the first time we’ve used light to control insulin release in a live animal,” Friedman says.
At present, the method requires ultraviolet light, which may prove damaging to human tissues over time. In future work, the team aims to adapt this technique to use visible light instead. They also plan to improve the polymer’s insulin-carrying capacity and make the material biodegradable. “Even with this first-generation material we could see enough insulin release to cause a significant decrease in blood glucose,” Friedman says. “In the long term, we want materials that leave no trace after the insulin is used up.”
The work is “a creative strategy for a really important problem,” says Ivan J. Dmochowski of the University of Pennsylvania, who was not involved with the study. He adds that the method could apply to many other small proteins or drugs that need to be administered at regular intervals. “It doesn’t completely get around the problem of needing to do injections, but offers more flexibility for drug delivery options,” he says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter