Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Hairy, expanding nanoparticles produce versatile coatings
Particles that change size with pH could help scientists fine-tune surface coatings
by Matt Davenport
December 9, 2016

A team of European scientists think about hairy polymer nanoparticles much like Cruella de Vil thought about furry Dalmation pups: Both would make fine coats. But chemistry, rather than cruelty, is helping these researchers achieve their goals.
Nanoparticle-based coatings help researchers tailor the optical and mechanical surface properties of the objects they cover. For example, researchers at Toyota Central R&D Laboratories recently showed that silica particles could help make flexible, antiglare coatings. Creating coatings with different surface properties, however, typically demands that researchers make different suspensions filled with different nanoparticles tailored for each new application.
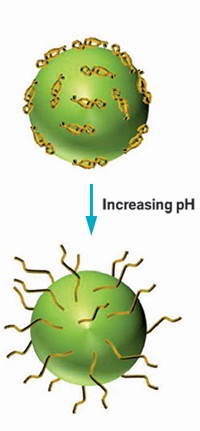
Joseph L. Keddie of the University of Surrey and his colleagues have now developed “hairy,” growing particles that could simplify that process. Working with chemists at France’s National Center for Scientific Research (CNRS) in Lyon, Keddie and his crew created spherical copolymer particles adorned with mops of poly(methacrylic acid) chains that extend with increasing pH (ACS Appl. Mater. Interfaces 2016, DOI: 10.1021/acsami.6b12015).
At low pH, the chains cling to a particle’s core, leading to spheres with a diameter of roughly 50 nm. But as the pH becomes basic and the chains extend, the nanoparticle’s diameter grows, reaching more than 90 nm above pH 7. By mixing these size-shifting particles with larger, fixed-size acrylate particles, the team created a variety of coating motifs.
For instance, at low pH, these two-particle suspensions dry to form layered coatings with smaller particles sitting atop the larger ones. But at basic pH values, particles are “jammed” into place as they dry, resulting in homogenous coatings, Keddie says. This variety of coating styles should allow researchers to use a single colloidal suspension to tune a variety of surface properties, such as hardness, reflectivity, and potentially even stickiness, the Surrey team says.
John G. Tsavalas, a polymer chemist at the University of New Hampshire, agrees. “This is the one stone, two birds, analogy where a single composition can be triggered to be suited to a wide variety of end use applications.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter