Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
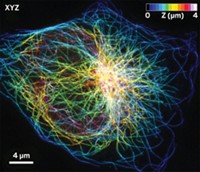
Researchers take superresolution microscopy to the next level
New method, dubbed MINFLUX, achieves nanometer resolution and fast analysis by combining the best of earlier imaging techniques
by Celia Henry Arnaud
December 22, 2016

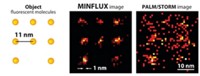
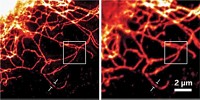
Existing superresolution fluorescence microscopy methods such as STED and PALM have trouble imaging molecules with spatial resolution better than 20 to 40 nm. A new superresolution method called MINFLUX combines aspects of those earlier methods to resolve molecules just 6 nm apart with nanometer precision. The method is fast enough for short- and long-range tracking of individual molecules. (Science 2016, DOI: 10.1126/science.aak9913).
STED and PALM (and the related technique STORM) distinguish densely packed features by allowing isolated molecules to fluoresce while those around them remain dark. The methods differ in how they determine the position of that fluorescence. In STED, a laser beam focused into a doughnut shape turns off fluorescence beneath the beam but not at the doughnut’s center. In PALM, fluorescent molecules are randomly switched on and off repeatedly so that only a few well-separated ones are activated at a time. The position of the activated molecules is determined with a camera.
“The strength of STED is that the doughnut always determines where the molecules are on and off and hence where the signal comes from,” says Stefan W. Hell of the Max Planck Institute for Biophysical Chemistry, who won the 2014 Nobel Prize in Chemistry for STED and leads the team that invented MINFLUX. On the other hand, “the strength of PALM is that you’re working with individual molecules.” But the disadvantage of PALM is that those fluorescent molecules must emit many thousands of photons in order for scientists to get enough fluorescence intensity to determine their positions precisely.
In the new method, Hell, Francisco Balzarotti, also at the Max Planck Institute for Biophysical Chemistry, and coworkers use a doughnut-shaped laser beam for exciting fluorescence instead of turning it off. Because there’s no light intensity in the doughnut hole, a molecule located entirely within the hole won’t fluoresce. But a molecule that’s even slightly offset from the hole does fluoresce. How much it fluoresces depends on how far it is from the beam’s zero-intensity center. By scanning the doughnut in a defined pattern over the sample, the researchers can determine the precise position of a molecule from how its fluorescence emission changes.
“Because we can relate the position of the molecule to the ‘zero’ of the doughnut, many fewer photons are required to find out the position of the molecule than in PALM,” Hell says.
The team used a molecular assembly strategy called DNA origami to construct two arrays of fluorescent molecules at defined distances from one another: 11 nm in one array and 6 nm in the other. MINFLUX determined the molecules’ positions with a precision of 2.1 nm and 1.2 nm, respectively.
In addition to improved spatial resolution, the MINFLUX measurements are fast enough and require such low laser intensity that the researchers used the method for tracking individual molecules in live cells at multiple time scales.
“It’s a big deal that this method can be used for imaging but also for tracking at short and long time scales,” says Christy F. Landes, a superresolution expert at Rice University. “Not all methods are useful for these three very different imaging applications.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter