Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.

Antibody-drug conjugates are promising next-generation cancer therapies that can target and selectively kill malignant cells while sparing healthy ones. These conjugates—in which a drug is bound to an antibody through a small chemical linker—harness the antibody’s ability to recognize markers specific to cancer cells and bring the potent drugs to their intended site of action. But one conundrum that has plagued drug developers and scientists is how to consistently tether a drug onto a predictable site on an antibody.
Now, chemists have reported a new metallopeptide catalyst that can reliably link drug molecules to a variety of antibodies, at the same location on the antibody every time (J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b06428). Inconsistent linkage can make it difficult for researchers to study the biological effects of these new compounds, or interfere with the antibody’s ability to bind to its target.
Previously, scientists used genetic engineering to create antibodies with chemical groups to which drugs could be attached. But this new approach bypasses the need for genetic engineering, a time-intensive process. The catalyst consistently binds to a particular location on an antibody’s so-called “constant chain,” an amino acid sequence that is found in all human antibodies and is conserved among many species. The catalyst produces reproducible results on human, pig, rabbit, and dog antibodies, the researchers found.
“Antibodies are pretty complicated molecules,” says Dennis G. Gillingham, a chemical biologist at the University of Basel not involved in this study. “This new technique is truly unique because it doesn’t require any engineering of any amino acids; the catalyst controls the reaction.”
Zachary T. Ball of Rice University and colleagues made the catalyst by taking a segment of a protein isolated from the bacterium Staphylococcus aureus that binds to the asparagine-79 residue on an antibody’s constant chain and then coordinated the peptide with three pairs of rhodium (II) ions. The rhodium complexes catalyze the addition of a linker group to the antibody—an alkyne-functionalized diazo that can react with many other small molecules, including drugs or fluorescent dyes, in a simple click-chemistry reaction.
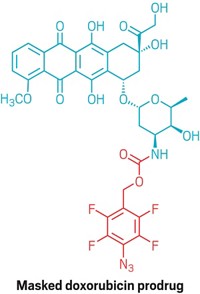
To prove that antibody-drug conjugates made with the catalyst can target cells, the authors attached fluorescent dyes and the chemotherapy drug doxorubicin to Herceptin, an antibody drug that recognizes the HER2 protein on the surface of mammalian breast cancer cells, and then visualized the localization using confocal microscopy. The dye appeared at the cell surface, confirming that the antibody still recognized the cell surface markers.
“We’re building next-generation drug conjugates using this method and testing those in cell and tumor model studies,” Ball says. He adds that it would be relatively easy to produce the catalyst in bulk for future drug development applications.
CORRECTION: This story was updated on Sept. 27, 2017, to correct the number of rhodium ions coordinated with the peptide.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter