Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.

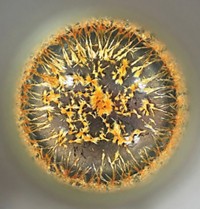
Under normal lighting conditions, this separatory funnel looked totally black. But when Lynn Stevens put it under an ultraviolet lamp, these distinct yellow and black phases revealed themselves. Stevens, an undergraduate at St. Mary’s College of Maryland, ran a reaction to make a fluorescent-yellow boron difluoride dye and was using the funnel to separate the product from unreacted starting material and other impurities. The dye dissolved into the organic dichloromethane phase, making it glow strongly yellow, while the less fluorescent impurities moved into the aqueous phase. Unfortunately, the separation got stuck in this jumbled emulsion. Stevens eventually broke the emulsion into two clean, separable layers by adding some sodium chloride to the mixture.
Do science. Take pictures. Win money. Enter our photo contest here.
For more Chemistry in Pictures, visit our new home on cen.acs.org.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter