Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Why baking soda could help boost the killing power of some antibiotics
Bicarbonate can disrupt the pH gradient across bacterial membranes and affect the potency of antibiotics
by Erika Gebel Berg
January 31, 2018
With antibiotic resistance on the rise, scientists are looking everywhere for better treatment approaches, even inside a box of baking soda—otherwise known as sodium bicarbonate. Bicarbonate ions, like the ones in this kitchen staple, act as a ubiquitous buffer in the human body. In a new study, scientists have figured out that bicarbonate diminishes the pH gradient across bacterial membranes, which can help usher some antibiotics in and keep some out (ACS Infect. Dis. 2017, DOI: 10.1021/acsinfecdis.7b00194). Using bicarbonate to create more realistic physiological conditions in the laboratory could help scientists develop new antibiotics, or adding bicarbonate to certain existing antibiotics could improve their effectiveness, the researchers say.
For the past 60 or so years, researchers and clinicians have been turning to a particular broth for growing bacteria—the classic Mueller-Hinton broth—but this medium for fast growth isn’t designed to mimic human physiology, says Eric D. Brown of McMaster University. A few years ago, he says, “we had an antibiotic compound that worked great in a mouse but had little or no activity in Mueller-Hinton broth.” Putting the antibiotic in a tissue culture medium, however, increased its activity in a dish, matching the animal results. The key ingredient—missing in the broth but present in the tissue culture medium—turned out to be bicarbonate.
To figure out the mechanism behind bicarbonate’s ability to alter antibiotic activity, the researchers looked at the impact of this buffer on the activity of a wide variety of antibiotics. They found that bicarbonate increased bacteria-killing for some drug classes and decreased it for others. The activity of many antibiotics is influenced by proton motive force, the product of cellular respiration that generates the energy molecule ATP. The large shifts in activity they observed were strong clues that bicarbonate was impacting this aspect of bacterial physiology, Brown says.
For example, bicarbonate decreased the potency of several tetracyclines. “The way tetracycline enters the cell depends on the pH gradient across the bacterial membrane,” Brown says. Protonation of tetracycline on the outside of the cell and deprotonation inside of the cell is thought to facilitate uptake and retention of the drug. Bicarbonate, however, decreases the pH gradient, thus reducing protonation of tetracycline and lowering uptake—an effect the researchers confirmed with fluorescence experiments.
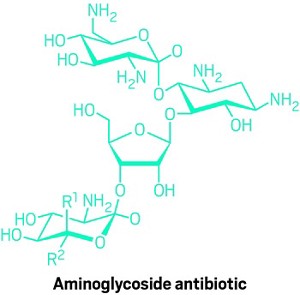
Conversely, bicarbonate increased the potency of many aminoglycoside antibiotics. The pH gradient is one driver behind proton motive force; the other is the charge differential across the membrane. If one driver is diminished, Brown says, the cell props up the other. So when bicarbonate decreases the pH gradient, which reduces the proton motive force, the cell compensates by increasing the charge differential. Aminoglycosides rely on the charge differential to enter cells, so an increase allows them to cross the bacterial membrane more effectively, explaining the boost in activity.
Brown’s team next looked at how bicarbonate impacted the body’s natural antibiotics, including antimicrobial peptides, which are chemical components of the innate immune system. Adding bicarbonate to the natural peptide indolicidin, for example, in standard growth media reduced the amount of peptide needed to halt the growth of Escherichia colifrom 100 µg/mL to just 1 µg/mL. “That means, if you use the right buffer, you can get a hundredfold increase in potency. That’s a real eye opener,” Brown says.
“I think this paper does a nice job proving that the biochemical mechanisms explain why you get different results,” says Victor Nizet of the University of California, San Diego. “Understanding the mechanism points to new therapies.” Indeed, Brown’s team is currently evaluating topical antibiotic ointments containing bicarbonate.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter