Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Energy
A practical membrane separates gasoline from crude oil
Stable nanoporous membrane could be made on large scale using established techniques
by Prachi Patel, special to C&EN
June 13, 2022

Separating the complex mix of hydrocarbons found in crude oil is crucial for producing fuels and chemicals. It’s done today using energy-intensive distillation. Now researchers have developed a potential alternative would save energy, demonstrating porous polymer membranes that can separate gasoline from light crude (Science 2022, DOI: 10.1126/science.abm7686).
Polymer membranes are key enablers of desalination and gas separation. For these applications, which otherwise require heating mixtures for separation, membranes have the advantage of low energy use and compact size. But polymer membranes have been challenging to adapt for use in separating petroleum products because they typically only work under mild conditions, says Suzana Nunes, a chemist at King Abdullah University of Science and Technology. Only one paper has been published on membrane-based crude oil separation (Science 2020, DOI: 10.1126/science.aba9806).
Separating crude oil and other complex chemicals mixtures using membranes requires diluting the mixtures in solvents and using temperatures of 60 to 90 °C to help transport the chemicals across the membranes. These conditions can damage the pore structure in polymer membranes. “We want to challenge membrane technology with separation applications that have harsh conditions,” she says.
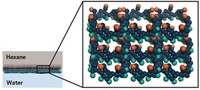
Nunes and her colleagues made membranes from polytriazole, a polymer that can withstand temperatures up to 100 °C, oxidation, and harsh solvents. Nunes first started working with this polymer 20 years ago for fuel cell membranes. Its backbone of triazole and fluorinated groups makes it extremely stable, she says, and it can be easily functionalized and made in large quantities. For crude oil separation, the team added dangling OH groups, which crosslink with each other when heated.
The membranes, which the researchers made using an established technique, have a layered structure, with an ultrathin layer of sub-nanometer sized pores at the surface and larger ones in the interior. The smaller pores at the surface exclude large molecules, and the larger pores inside allow liquid to rapidly flow through the membrane.
By using different solvents and processing temperatures and times to make the membranes, the researchers can tune pore sizes to selectively filter different hydrocarbons. “We could in principle have a series of membranes to get fractionation,” Nunes says. In experiments with light crude oil—which is mainly a mix of gasoline, kerosene, and diesel—the membrane allowed 80–95% of gasoline-sized hydrocarbon molecules to pass through.
The researchers now plan to make the membranes on flexible substrates such as carbon cloth. That would allow large-scale production using a roll-to-roll process.
Ryan P. Lively, a chemical engineer at the Georgia Institute of Technology, says that these nanoporous membranes show impressive selectivity and permeability. The polymer and fabrication process that the researchers chose—dissolving the polymer in a solvent, casting it, and immersing it in water before a heat treatment—also make it practical. Lively published the first paper on membrane-based crude oil fractionation, and he says there are many questions left to answer before this technology can find practical application.
“The main challenge facing membranes is the enormous scale of crude oil processing,” Lively says. Replacing well-established processes like distillation with membranes likely won’t happen quickly. Instead, a gradual change, say by integrating membranes with existing systems might be “one way to introduce the benefits of membranes into the petrochemical refinery,” he says, “but more experiments, process analysis, and modeling are needed.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter