Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Inorganic Chemistry
Structure of Pepto-Bismol active ingredient solved
Bismuth subsalicylate has layered structure
by Celia Henry Arnaud
April 26, 2022

People have taken bismuth subsalicylate to treat upset stomachs and nausea for more than a century. The compound is the active ingredient in the bright pink concoction sold as Pepto-Bismol. Despite that long history of use, bismuth subsalicylate’s structure has remained a mystery—one that researchers have now solved.
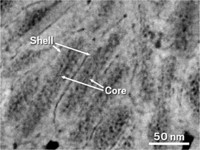
A. Ken Inge, Tom Willhammar, and coworkers at Stockholm University used a combination of electron diffraction and electron microscopy to solve the structure (Nat. Commun. 2022, DOI: 10.1038/s41467-022-29566-0). They found that the compound has a layered structure with variations in the stacking. The heterogeneity caused by the variations helps explain why the structure has been so hard to pin down.
The degree of disorder in the structures differed depending on the source of the bismuth subsalicylate. Compounds filtered out of Pepto-Bismol were too disordered to get a clear structure. But the researchers could obtain a structure by electron diffraction using material purchased from Sigma-Aldrich.
When the chemists analyzed the material with scanning transmission electron microscopy, they could finally see the packing of the subunits in the layers. “Although we found pretty large ordered domains, we also saw domains of the crystal where we had imperfections in the ordering,” Willhammar says. “We could see that this was the source of why it was so challenging to determine the structure originally.”
The new structure shows that O2− anions bridge Bi3+ cations in a rodlike structure. Along the structure, the O2− anions alternatingly connect three and four Bi3+ cations. Some of the salicylate anions coordinate to Bi3+ through only their carboxylate group; others coordinate to Bi3+ through both the carboxylate and phenolic groups.
“The results can potentially shed some new light on the mechanism of action of [bismuth subsalicylate],” says Andrea M. Goforth, who studies bismuth nanoparticles at Portland State University. “Since the structure is now known, we can see how the hydrophilic and hydrophobic parts spatially distribute and have a better picture of the intermolecular forces and structural disorder.” The intermolecular forces can affect properties such as solubility and bioavailability, she says.
The Stockholm team plans to dig deeper into the structure of bismuth subsalicylate. “I’m curious, if you have more disorder, how that affects the physical properties of the material,” Inge says. In addition, the researchers plan to use their methods to determine the structures of other bismuth-based drugs whose structures aren’t yet known.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter