Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Polymers
Hydrogel stays strong and flexible even at freezing temperatures
The freeze-proof, self-healing hydrogel could expand the temperature range for some electronics and medical devices
by Prachi Patel, special to C&EN
June 26, 2019

Squishy-yet-strong hydrogels are invaluable for making medical devices, tissue engineering scaffolds, and sensors. Being mostly water, though, they freeze and become useless in subzero temperatures. But now, researchers have made a freezable hydrogel that boasts high strength, moldability, adhesiveness, and self-healing powers (ACS Appl. Mater. Interfaces2019, DOI: 10.1021/acsami.9b05652).
The new hydrogel stays stretchy down to –80°C and is stronger than most other synthetic and natural polymer-based gels, says Junjie Li, a chemical engineer at Tianjin University. Its combination of mechanical properties should open up applications in flexible energy-storage devices, soft electronics, and wearable devices designed for subzero conditions. The hydrogels could also be used to protect cells, tissues, and organs stored at cryogenic temperatures from cold damage.

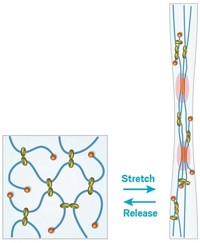
Hydrogels are three-dimensional networks of cross-linked polymer chains dispersed in water. When frozen, hydrogels lose their elasticity and become brittle. To make a freeze-tolerant hydrogel, Li, Fanglian Yao of Tianjin University, and their colleagues replaced part of the water in their gelatin-based hydrogel with glycerol. The simple one-step synthesis involves soaking a gelatin hydrogel precursor in a water-glycerol solution containing sodium citrate for 3 h.
“Glycerol forms hydrogen bonds with water molecules, which can effectively prevent freezing of water at subzero temperatures,” Yao says. The strength of the gel comes from hydrogen bonds between the gelatin chains and glycerol and from ionic interactions between the ammonium groups in gelatin and the citrate ions. The gelatin chains also easily intertwine and untwine, making the gel moldable and healable.
To show off the hydrogel’s properties, the researchers formed it into various shapes and stretched it to at least five times its original length at room temperature. It remained transparent and the researchers could still stretch and twist it after freezing to –80°C for three days, whereas a traditional hydrogel shrunk and became brittle and opaque under the same treatment.
When the researchers pressed two pieces of their hydrogel together and heated it for a half hour, the pieces fused into one that retained about half of the material’s original strength. And when warmed and pressed against metal, glass, and plastic, the gel stuck fast, without any glue.
Others have added salt to hydrogels to make them freeze tolerant. But those previous hydrogels have not been strong or have required sophisticated manufacturing techniques, Li says.
The gel’s outstanding properties and easy production method should make it important for practical applications, says Mingjie Liu, a chemist at Beihang University. The only downside he sees is that the results might not work with other hydrogel chemistries. “The mechanical strength enhancement based on the introducing of citrate might not work for all hydrogels or polymers,” he says.
CORRECTION
This story was updated on July 1, 2019, to correct the type of chemical groups on the gelatin chains. They are ammonium groups, not ammonia groups.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter