Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Computational Chemistry
Simulations suggest graphene’s elusive cousin may become a reality
Simulations show zeolites can be templates to make hypothesized Schwarzite energy-storing materials
by Sam Lemonick
August 15, 2018
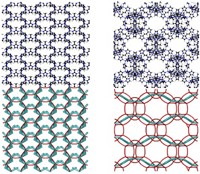
The discovery of graphene and fullerenes like carbon nanotubes kicked off an ongoing rush to put those materials to work. Now there’s reason to think the hypothesized third member of that family might soon join them in the real world (Proc. Natl. Acad. Sci. USA 2018, DOI: 10.1073/pnas.1805062115).

Geometrically, graphene has zero curvature while fullerene allotropes such as buckyballs and carbon nanotubes are positively curved. The negatively curved allotropes—think of the shape of an hourglass—are known as Schwarzites. Schwarzites’ hypothesized electronic and mechanical properties could make them useful in energy storage, gas capture, or catalysis. Yet attempts to make Schwarzites experimentally have come up short so far. One promising avenue has been to use porous aluminosilicate minerals called zeolites as a template. Carbon atoms deposited on the interior surfaces of zeolites can form continuous shapes resembling Schwarzites, though the real thing has remained elusive.
Now, Berend Smit’s laboratories at the Ecole Polytechnique Fédérale de Lausanne (EPFL) and the University of California, Berkeley, have developed a computational method which suggests that some kinds of zeolite-templated carbons (ZTCs), including some that have been attempted in labs, are in fact Schwarzites. Smit credits the project’s success to collaboration between chemists and mathematicians in his group. They simulated lining hundreds of available computer models of known and theorized ZTCs with carbon atoms. The researchers mathematically compared the surfaces of the resulting ZTCs to those of Schwarzites and found 15 matches. “I think the race is on now from the experimental side,” says Efrem Braun, a graduate student in Smit’s laboratory whose interest in ZTCs started this research project.
Chemists have already come close with three of the ZTCs this study identified as Schwarzites. “Rigorously speaking, all actually synthesized ZTCs so far, including my samples, are not yet perfectly Schwarzites,” says Ryong Ryoo, a chemist at Korea Advanced Institute of Science & Technology who studies zeolites and other mesoporous materials. Ryoo says ZTCs he and others have made contain framework defects.
Humberto Terrones of Rensselaer Polytechnic Institute says the new paper “will get experimentalists going.” Terrones and Alan Lindsay Mackay of the University of London theorized Schwarzites’ existence in 1991. Terrones adds the research “reinforces the idea that impregnating zeolites with a carbon source may generate Schwarzites.” But he says experimentalists still have work to do to find the right conditions to synthesize pure Schwarzites, which would contain only sp2-hybridized carbons and no defects.
If Schwarzites can be made, Smit says their conductivity and porosity may make them excellent supercapacitors, which can store energy for short periods and release it very quickly. He says preliminary calculations show promise but only actual Schwarzites can provide concrete evidence.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter