Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Germanium gets chiral
A mild carbene insertion reaction imparts stereochemistry to the main-group element
by XiaoZhi Lim, special to C&EN
February 9, 2024

Like carbon and silicon, germanium’s lighter brethren on the periodic table, the metalloid can bond to four different groups, forming a chiral, tetrahedral molecule. Researchers at Nankai University led by Qi-Lin Zhou have devised a way to make these rare germanium compounds, reporting dozens of molecules with stereocenters of that element (J. Am. Chem. Soc. 2024, DOI: 10.1021/jacs.3c14386).
Although Zhou’s group prepared a variety of heteroatom chiral compounds in the past, including those of silicon, phosphorus, and boron, chiral germanium compounds have remained elusive; just one example had ever been reported. So “curiosity was our main driving force,” Zhou says.
Germanium’s slipperiness can be partly attributed to its larger size and empty 3d orbitals, which have a penchant to bond to five or six groups rather than the four found at tetrahedral stereocenters. Also, because germanium’s bonds with organic groups are often weaker than lighter heteroatoms’, the groups are more likely to pop off and on in solution, scrambling their arrangement and making it hard to capture a set stereochemistry.
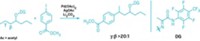
To make the new compounds, the researchers designed a chiral rhodium-based catalyst and used it to react a carbene with a germanium molecule sporting two germanium-hydrogen bonds. At room temperature and pressure, the carbene inserted itself into one of the Ge–H bonds, creating a new chiral compound.
The researchers used the reaction to prepare dozens of different molecules. By simply lengthening the reaction time, the researchers scaled up the preparation of one compound from tens of milligrams to more than a gram. They also managed to further react the remaining Ge–H bond in that compound without disrupting the compound’s stereochemistry.
The researchers plan to fully investigate the properties of these chiral germanium compounds, Zhou says. The metalloid has semiconducting properties, which makes him hopeful that its compounds could lead to novel materials for semiconductor or electronic applications, he says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter