Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Reaction Mechanisms
Mechanochemistry strips cargo molecules from a loaded rotaxane
Using polymer strands to pull a ring along an axle could release repair molecules in self-healing materials
by Mark Peplow, special to C&EN
April 11, 2024
Do you remember the simple childhood pleasure of sliding a pinched thumb and forefinger along a stalk of meadow grass to strip its seeds and toss them into the air? Researchers have now re-created this joyful experience at the nanoscale, offering a method that might be used to deliver drugs or release molecules that repair damaged materials (Nature 2024, DOI: 10.1038/s41586-024-07154-0).

The system relies on mechanochemistry, in which a mechanical force causes a chemical reaction. One way to deploy this force is through polymer mechanochemistry, which involves tethering long polymer strands to a force-sensitive molecule known as a mechanophore. “The idea is to use polymers as a rope in a tug-of-war,” says Guillaume De Bo of the University of Manchester, who led the new work.
In solids, an impact can tug the polymer strands apart and trigger the mechanophore’s reaction. Researchers have previously used this strategy to release fluorescent molecules to signal damage in a material. In solution, bursts of ultrasound have the same mechanochemical effect. But each tug-of-war bout can free only a single molecule, which limits the concentration of released molecules.
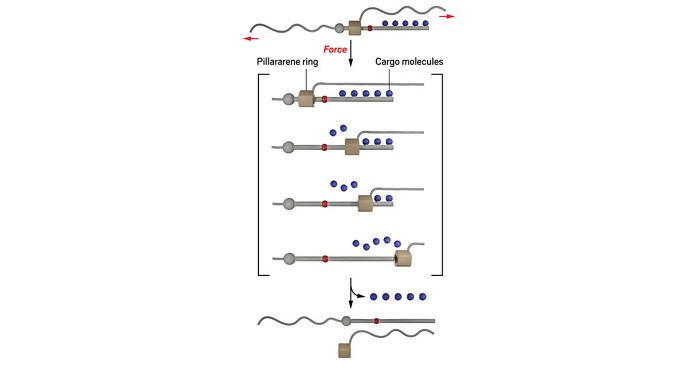
De Bo’s team has now overcome this drawback with the help of a rotaxane, a structure containing a molecular ring threaded onto an axle-like structure. The long axle contains up to five furan groups that act as docking stations so that maleimide-based cargo molecules can be added by a Diels-Alder reaction. The molecular ring, a pillararene, is fitted with a long strand of polymethyl methacrylate that runs roughly parallel to the axle. Another polymer strand is attached to one end of the axle so that it can pull in the opposite direction.
When ultrasound pulses tug these two polymers apart, theirmovement hauls the pillararene along the axle so hard that it triggers a reverse Diels-Alder reaction at each docking station, stripping off all the cargo molecules. “Being able to release multiple molecules in a single elongation event—this is completely new in mechanochemistry,” De Bo says.
For rotaxanes with a single docking station, ultrasound freed up to 71% of the maleimide cargo units in solution, while compression of a bulk sample released 30%. Although rotaxanes with five loaded docking stations released a lower percentage of their cargo molecules, they released more molecules in total. “This is already a very good efficiency,” says Anne-Sophie Duwez, an expert in single-molecule force spectroscopy at the University of Liège, who peer-reviewed the paper. “It’s a very elegant system. They really had to master different aspects of chemistry to reach their goal.”
The researchers then showed that the system also worked with three useful cargo molecules—a fluorescent tag, a catalyst, and the anticancer agent doxorubicin. This proof of principle suggests that the method might be used to build an ultrasound-activated drug delivery system.
For now, De Bo’s team is creating rotaxanes with more docking stations, and it also hopes to load the rotaxane’s axle with a mixture of monomers and a catalyst. In principle, a sudden force could release these ingredients to generate new polymers that repair a damaged region of material. “The ultimate goal would be to create an autonomous self-healing system whereby the mechanical damage triggers the release of healing agents that would reform the material,” De Bo says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter