Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
ACS Meeting News
Extremophile enzyme links lipid tails
Iron-sulfur clusters are key to coupling unactivated carbons
by Bethany Halford
August 25, 2022
| A version of this story appeared in
Volume 100, Issue 30
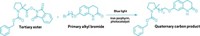
In pursuit of making molecules, chemists have sought ways to build bonds between unactivated sp3-hybridized carbons. Although there are chemical catalysts that can accomplish this feat, this kind of coupling was unknown in natural systems. Scientists now report that an enzyme from the extremophile Methanocaldococcus jannaschii connects unactivated sp3-hybridized carbons at the ends of two lipid tails to create large macrocyclic ethers. M. jannaschii lives in ocean vents, where there are extreme temperatures and pressures, and these large ethers add stability to the extremophile’s membrane.
How the enzyme manages to make this connection was a mystery, according to Pennsylvania State University’s Squire J. Booker, who presented the work at the ACS Fall 2022 meeting on Wednesday, during a talk for the Division of Inorganic Chemistry. Because of the two unactivated carbons at the ends of the lipids, the enzyme must do radical chemistry, Booker said. Once you’ve generated the first radical, “how do you store that radical long enough to be able to generate a second radical to allow them to couple?” he asked.
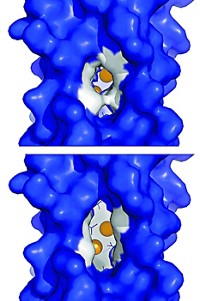
Cody T. Lloyd, a graduate student in Booker’s lab, worked in collaboration with Penn State’s Amie K. Boal to determine the X-ray crystal structure of the enzyme. When the researchers looked at its electron density, they saw a hydrophobic tunnel that terminated near an iron-sulfur cluster in the enzyme’s active site. That suggested that the lipid tails were reaching into that portion of the enzyme, and X-ray crystal structures of the enzyme and the lipid substrate showed this to be the case.
The researchers think that the enzyme stores the radical it generates by forming a covalent bond between the end of the lipid tail and a sulfur in one of the enzyme’s iron-sulfur clusters. When the enzyme generates a second substrate radical, “that radical attacks the carbon that’s bonded to the sulfur of the iron-sulfur cluster. And that’s how you make your carbon-carbon bond,” Booker said. “That was completely unexpected and completely new.” The work was also published in Nature (2022, DOI: 10.1038/s41586-022-05120-2).
Daniel L. M. Suess, who studies iron-sulfur clusters at the Massachusetts Institute of Technology, said in an email that this is “a critical and surprising role for iron-sulfur clusters.” The finding, Suess added, raises other questions about the function of iron-sulfur clusters in enzymes: What governs the stability of sulfur-carbon bonds when they’re part of an iron-sulfur cluster? Why does the carbon form a bond with sulfur instead of iron? “Answering these and related questions will further our understanding of the rich reaction chemistry performed by iron-sulfur cluster enzymes,” he said.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter