Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Science Concentrates
July 5, 2004
| A version of this story appeared in
Volume 82, Issue 27
A hormone analog boosts sexual desire in female rats [Proc. Natl. Acad. Sci. USA, published online June 28, http://dx.doi.org/10.1073/pnas.0400491101]. Researchers first noticed that PT-141, a cyclic heptapeptide that mimics the action of a-melanocyte-stimulating hormone (-MSH), causes erections in men. But James G. Pfaus of Concordia University, in Montreal, wondered whether the neuropeptide also affects females. He and his team observed that female rats on PT-141 initiate sex more by hopping, darting, and enticing males to chase them. This is the first time that a melanocortin receptor has been linked to sexual desire, Pfaus says. Unlike other erectile enhancers such as Viagra, which relaxes vascular smooth-muscle cells and increases blood flow to the penis, PT-141 targets receptors in the brain:  -MSH receptors 3 and 4. The finding could lead to a new drug for women, who currently have no pill for sexual disorders.
-MSH receptors 3 and 4. The finding could lead to a new drug for women, who currently have no pill for sexual disorders.
By systematically substituting esters for amides in the peptide backbone of a small protein, researchers have managed to obtain a remarkably detailed picture of its folding [Nature, 430, 101 (2004)]. The role of backbone hydrogen bonds—those between one amide’s NH and another’s carbonyl oxygen—in protein folding remains controversial because the peptide backbone cannot be mutated by conventional methods. Jeffery W. Kelly of Scripps Research Institute and coworkers synthesized a series of protected, chiral  -hydroxy amino acid analogs and used them to make 19 different amide-to-ester mutants of a small
-hydroxy amino acid analogs and used them to make 19 different amide-to-ester mutants of a small  -sheet protein via solid-phase chemical synthesis. They find that the five hydrogen bonds (red dashed lines) in the protein’s hydrophobic core are crucial to the folded protein’s thermodynamic stability. Hydrogen bonds in a solvent-exposed loop, however, determine the rate of protein folding. When combined with side chain mutagenesis data, amide-to-ester mutations will allow protein folding to be charted at the atomic level, the researchers say.
-sheet protein via solid-phase chemical synthesis. They find that the five hydrogen bonds (red dashed lines) in the protein’s hydrophobic core are crucial to the folded protein’s thermodynamic stability. Hydrogen bonds in a solvent-exposed loop, however, determine the rate of protein folding. When combined with side chain mutagenesis data, amide-to-ester mutations will allow protein folding to be charted at the atomic level, the researchers say.
Mass spectrometric analysis of surface-bound organic and biomolecular species is often hampered by the failure to form ions that are structurally and quantitatively representative of those species. Such analyses are applicable to arrays used in chemical sensing and diagnostics. A team led by Luke Hanley of the University of Illinois, Chicago, uses single-photon ionization (SPI) to circumvent some of those difficulties. They use the Fmoc protecting group—N- -(9-fluorenylmethyloxycarbonyl)—as a chemical tag that allows SPI of neutral molecules that have been desorbed from the surface [Anal. Chem., published online June 24, http://dx.doi.org/10.1021/ac049434t]. The chemical tag can be photoionized with a fluorine laser—which has too low a photon energy to ionize many organic species—with only minimal fragmentation of the tethered molecule. SPI can be combined with any desorption method and allows quantitative analysis.
-(9-fluorenylmethyloxycarbonyl)—as a chemical tag that allows SPI of neutral molecules that have been desorbed from the surface [Anal. Chem., published online June 24, http://dx.doi.org/10.1021/ac049434t]. The chemical tag can be photoionized with a fluorine laser—which has too low a photon energy to ionize many organic species—with only minimal fragmentation of the tethered molecule. SPI can be combined with any desorption method and allows quantitative analysis.
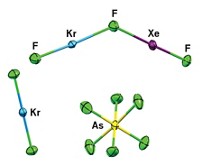
More than 40 years ago, chemist Neil Bartlett and others burst the myth that the “inert gases” were chemically inert by synthesizing xenon compounds. In later work, Bartlett and others showed that XeF2 can act as a ligand for metal ions, but the metal centers were simultaneously coordinated by AF6– (A = As, Sb, P). Boris emva of Joef Stefan Institute, in Ljubljana, Slovenia, and coworkers have now prepared the first compound in which a metal center is coordinated only to XeF2 molecules [Angew. Chem. Int. Ed., 43, 3456 (2004)]. The researchers synthesized Ca2(XeF2)9(AsF6)4, a white crystalline solid, by reacting a 20-fold excess of XeF2 with Ca(AsF6)2 in anhydrous HF solvent. The two calcium atoms (white) are crystallographically different: One calcium atom is coordinated to fluorine atoms from the four AsF6– ions and from four XeF2 molecules, while the other calcium atom is coordinated to fluorine atoms from all nine XeF2 molecules (As = blue, F = green, Xe = pink). The team hopes next to synthesize a compound where all metal centers are coordinated only to XeF2.
Anaerobic metal-reducing bacteria have been shown to play a key role in releasing arsenic from their host sediments into groundwater. The discovery may help in the development of strategies for reducing the widespread arsenic contamination of water wells in West Bengal, India, and in Bangladesh. In a laboratory-based study, microbiologists Farhana S. Islam and Jonathan R. Lloyd at the University of Manchester, in England, and their coworkers mixed sediments collected from a contaminated aquifer in West Bengal with simulated groundwater and incubated the mixtures at 20 °C under a range of biogeochemical conditions [Nature, 430, 68 (2004)]. Using liquid chromatography and inductively coupled plasma-mass spectrometry, the team showed that naturally occurring organisms in the sediments reduce Fe(III) to Fe(II) and then As(V) to As(III), which is more toxic and mobile than As(V). The organisms that catalyze the reductive mobilization of arsenic remain to be identified.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter