Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Prussian Blue Still a Hot Topic
Tricentennial of inorganic pigment celebrated with broad discussions on metal-cyanide chemistry
by STEPHEN K. RITTER, C&EN WASHINGTON
May 2, 2005
| A version of this story appeared in
Volume 83, Issue 18

Three hundred years ago, adventurous scientists working in the fledgling discipline of chemistry often weren't sure what they would get when cooking up a recipe. That was how Prussian blue was born.
This quintessential inorganic pigment, later identified as ferric ferrocyanide, Fe4[Fe(CN)6]3, was first synthesized by a German artist in 1704 as he attempted to make a red pigment. Never mind about the red, because Prussian blue quickly became a highly desired substance and remained in strong demand until the 1970s. Beyond its use as a pigment, Prussian blue is perhaps the most studied compound in chemistrydom.
Even after three centuries, it's still a hot material, as judged by the diversity of new research presented during a Division of Inorganic Chemistry symposium held at the American Chemical Society's national meeting in San Diego in March. Titled "The Metal-Cyanide Renaissance, On the Tricentennial of the Synthesis of Prussian Blue," the symposium focused primarily on the study and use of the compound and its analogs as molecule-based magnets.
The symposium's title was inspired by a review article on metal-cyanide chemistry coauthored in 1997 by symposium co-organizer Kim R. Dunbar, a chemistry professor at Texas A&M University. "Little did I know that the article would quickly become outdated, especially in the area of molecular magnets based on cyanide precursors," Dunbar said. "There's every reason to believe that the current trend of increasing activity in this field will continue, given the discovery of fascinating new multiproperty effects in cyanide materials, such as light-induced and pressure-induced magnetic behavior."
The idea for the Prussian blue symposium originated with State University of New York, Stony Brook, chemistry professor Stephen A. Koch, who helped organize the symposium but was unable to attend, Dunbar said. Koch began working on iron-cyanide chemistry in the late 1990s after it was reported that iron-nickel and iron-iron hydrogenase enzymes contained active sites with [Fe(CN)x(CO)y] coordination centers. Koch's group has synthesized new members of this compound family to study as models for catalysts and as two-dimensional polymeric materials.
Koch was aware that Prussian blue was an old compound, but initially he didn't know about its importance in the history of chemistry. "When you start working in a new area of chemistry, one of the things that you do is read the literature," Koch told C&EN. "It's rare that you have to go back 300 years."

When Koch realized that the Prussian blue tricentennial was approaching, he worked with Dunbar and co-organizer Jeffrey R. Long, an associate chemistry professor at the University of California, Berkeley, to put together the symposium. The goal was to bring together an international collection of researchers "to tell a story about metal-cyanide chemistry," Dunbar said.
APPROPRIATELY, the symposium began with a discussion of the history of Prussian blue by emeritus chemistry professor Andreas Ludi of the University of Bern, in Switzerland. "It's nice to be in front of an audience of cyanide fans," Ludi began, a comment that drew chuckles from the audience because it hinted at the toxicity of cyanide and its use for sinister purposes. Ludi, who worked on Prussian blue, then regaled attendees with the story of the original synthesis of Prussian blue and provided an overview of its chemistry.
Ludi likes to use the metaphorical label "inorganic evergreen" for Prussian blue because of its perpetual attraction for chemists. "Prussian blue has always been a part of inorganic chemistry," Ludi commented. "It was the first synthesized coordination compound. Prussian blue started as a very simple material as a pigment, gradually grew in to a compound that helped solve scientific challenges, and nowadays has become of interest for nanomaterials and molecular-magnet applications."
Because of wars in Europe at the turn of the 18th century, natural pigments had become hard to obtain and were expensive, Ludi explained. That led color makers to begin experimenting with ways to make pigments. One of these color makers was a German reportedly named Heinrich Diesbach, who created Prussian blue by chance in Berlin in 1704 while mixing up a "strange brew" that included ingredients such as dried ox blood, Ludi said.
Other key ingredients were potash (a mix of KOH, K2CO3, and other potassium salts) and green vitriol (FeSO4). Diesbach reportedly worked in association with an alchemist named Johann C. Dippel, who may have supplied Diesbach with some contaminated potash. Diesbach wasn't able to explain what happened during his formative reaction, but these days it's believed that the potash reacted with hemoglobin in the ox blood to form potassium ferrocyanide, K4Fe(CN)6. The reaction with iron sulfate in turn created ferric ferrocyanide, Fe4[Fe(CN)6]3, or Prussian blue.
This synthesis suddenly made available an affordable blue pigment, Ludi said. By 1710, it was already becoming a hit. The synthesis was published in England in 1724 in the Philosophical Transactions of the Royal Society, although without reference to Diesbach and Dippel. "It may have been a case of industrial espionage," Ludi indicated.
Major industrial production of the pigment began in 1785, more than 70 years before the production of the first organic dyes (C&EN, July 12, 2004, page 31). Most of the key historical figures of chemistry have used or studied Prussian blue at some point. It became widely used in inks, as laundry bluing, as a color additive for fertilizers, and as a paint pigment, Ludi added. Prussian blue was used in an early type of photography known as cyanotype and in making blueprints, carbon paper, and typewriter ribbons.
Prussian blue's "scientific career" began in 1936, when J. F. Keggin and F. D. Miles recorded the X-ray powder diffraction pattern and proposed a general structural model, Ludi noted. "Then after World War II came many new and fantastic physical tools, and Prussian blue was always the guinea pig for these techniques."
By about 1970, Ludi and his coworkers could grow single crystals of Prussian blue and its analogs and determine their crystal structures. The structure is a three-dimensional coordination polymer framework with Fe(III) and Fe(II) ions residing at the corners of cubes and linked by bridging cyanide ligands, Fe(III)–N–C–Fe(II). Water molecules reside in the interstitial spaces of the framework and are coordinated to the Fe(III) atoms since some of the sites for the cyanide groups and Fe(II) atoms are vacant, as indicated by the 4:3 ratio of the iron atoms.
There are a variety of possible other molecular shapes for metal-cyanide complexes besides the Prussian blue network structure. For example, Hiroki Oshio of the University of Tsukuba, in Japan, described cyanide-bridged "molecular squares" in his lecture in San Diego. These compounds are planar clusters with four metal atoms at the corners of a square joined together by bridging cyanide groups; additional ligands external to the square are required. These are the smallest building blocks for Prussian blue analogs, he noted. Other structural types include single-molecule magnets, where some of the metal coordination sites are blocked by single or multidentate ligands, leading to modest-sized metal clusters that individually exhibit the properties of a magnet below about 4 K.
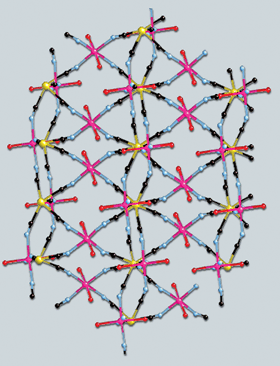
THE INTENSE BLUE color of Prussian blue arises from an electronic transition from the Fe(II) atoms to the Fe(III) atoms, Ludi noted. Prussian blue also is a ferromagnet below 5.5 K, another property resulting from the Fe(II)-Fe(III) electronic interaction.
Industrial synthesis of Prussian blue typically involves the reaction of Na4Fe(CN)6 and FeSO4 in the presence of (NH4)2SO4, followed by oxidation with a chromate or chlorate salt, Ludi said. Production peaked at about 50,000 metric tons per year in the mid-1970s, he noted, and since then the pigment gradually has been replaced by better performing materials, such as phthalocyanine blue.
"Prussian blue has a great history: It has a past, it has a present, and I am sure it will have a future," Ludi concluded.
Following that introduction, 30 invited speakers discussed the current chemistry of Prussian blue and its analogs, which include a variety of transition metals and bridging ligands. Some of the topics covered included host-guest chemistry; chemical synthesis and materials processing; catalysis; and the effects of temperature, light, and pressure on the magnetic properties.
Several speakers alluded to the fact that Prussian blue's open framework allows it to trap molecules or individual ions. Exploiting this capability, the Food & Drug Administration recently approved Prussian blue as a treatment for exposure to radioactive cesium that might occur following a terrorist attack using a "dirty bomb."
In a similar vein, Shin-ichi Ohkoshi of the University of Tokyo noted that a cobalt-manganese hexacyanochromate compound could serve as a magnetic humidity sensor. The material can reversibly adsorb and desorb water molecules as ligands for the cobalt atoms. This process causes a change in the coordination geometry of cobalt that switches the magnetic behavior on and off.
Andrew B. Bocarsly of Princeton University described preparation of microporous solid materials he calls cyanogels that are made from palladium-cobalt cyanide complexes using a sol-gel process. The cyanogels can selectively and reversibly adsorb CO2, he said.
Steven S. Kaye in Long's group at UC Berkeley reported that dehydrated M3[Co(CN)6]2 complexes, where M = Mn, Fe, Co, Ni, Cu, or Zn, can bind hydrogen molecules at exposed metal coordination sites (J. Am. Chem. Soc. 2005, 127, 6506). The copper-containing framework achieved the highest H2 loading at 1.8 weight %, Kaye said, which is slightly better than other reported metal-organic frameworks. It's still unclear if the materials can be optimized for bulk hydrogen storage, he added.
THE MAJORITY of the "metal-cyanide renaissance," apparent from the exponential growth of journal papers on Prussian blue and its analogs since 1990, is attributed to studies of the compounds' magnetic properties.
One speaker on this topic was Joel S. Miller of the University of Utah. Miller is considered a pioneer in the field of molecule-based magnets for his work on transition-metal tetracyanoethylene complexes and the study of new magnetic effects. In San Diego, Miller described recent chemistry in his lab to make [Ru2(O2CCH3)4]3[M(CN)6], where M = Cr, Co, or Fe, that underscores the differences in using aqueous versus nonaqueous solvent systems in cyanide chemistry.
When prepared in acetonitrile, the complex forms the expected 3-D network lattice structure typical of Prussian blue, he said. But when prepared in water, the crystal structure is made up of two identical lattices, one inside the void spaces of the other (Angew. Chem. Int. Ed. 2005, 44, 2416).
"Interpenetrating lattices have an architectural appeal for chemists, but only in rare cases are the resultant physical properties dramatically different," Miller told C&EN. He believes the interpenetrating version of the new compound may be the first good example of this phenomenon. The magnetic effects, besides being a previously unobserved anomaly for molecular magnets in general, differ from those of the noninterpenetrating lattice material.
Another intriguing complex Miller's group has uncovered contains a chromium(II) cyanide anion, [Cr(CN)5]3–, which has the highest electron spin state (S = 2) reported for a metallocyanide species (Angew. Chem. Int. Ed., published online April 14, dx.doi.org/10.1002/anie.200462763). Miller's group was attempting to make [Cr(CN)6]4– to use as a ligand in a new family of molecular magnets, and the researchers reacted excess [N(CH2CH3)4][CN] with Cr2(CO2CH3)4 in acetonitrile, he explained. But because high-spin [Cr(CN)6]4– would have an electron in an antibonding orbital, he thinks that the lower stability favors loss of cyanide and formation of [Cr(CN)5]3–.
Advertisement
Discovering this species helps to dispel the myth that cyanide anion is always a strong field ligand (influencing electron pairing in transition-metal-complex molecular orbitals) and nearly exclusively forms low-spin transition-metal complexes--an important finding for inorganic chemists, Miller told C&EN. The high-spin [Cr(CN)5]3– complex has a significantly higher magnetic susceptibility than the low-spin [Cr(CN)6]4– complex, as expected, he said.
Researchers in molecular magnetism are interested in studying the behavior of nanomaterial particles sized between the smallest possible metal or metal oxide particles and single molecules. Cobalt and other metals used for high-density data-storage devices can be processed to have particle sizes as small as about 3 nm in diameter. Particles of this size contain about 1,000 metal atoms and have a relatively high spin ground state (large number of unpaired electrons) of 1,000 or more that gives rise to magnetic properties. Single-molecule magnets, on the other hand, typically have only a few metal centers, fewer than 50 unpaired electrons, and a spin state of about 10 or below.
Laure Catala of the University of Paris South, in France, described new processing systems to control the size of metal-cyanide nanoparticles and their distribution in substrate materials. One system she presented was the use of reverse micelles to make Ni3[Cr(CN)6]2 nanoparticles.
Catala, Talal Mallah, and their coworkers prepared stable microemulsions [water/heptane/sodium bis(2-ethylhexyl)sulfosuccinate] of K3Cr(CN)6 and NiCl2 and mixed them together to create a single microemulsion. The Ni3[Cr(CN)6]2 nanoparticles, with an average size of about 3 nm, gradually formed on the interior of the micelles, she explained.
This reverse-micelle process has been used by another group to make nanoparticles of other Prussian blue analogs in the range of 10 to 50 nm, Catala said, but those researchers didn't report the isolation of the nanoparticles from the microemulsions, which prevented characterization of the magnetic properties. Catala and her colleagues were able to separate the individual particles by adding p-nitrobenzylpyridine to the microemulsion. The substituted pyridine molecules coordinate to the nickel atoms on the nanoparticle surface and allow the particles to be recovered and then dispersed in an organic solvent for magnetic characterization. The particles also can be isolated in a poly(vinylpyrrolidone) matrix and characterized, she said.
The ground spin state of the nanoparticles is about 130, ideally intermediate between metal particles and single-molecule magnets, Catala related. "They are the first clear example of superparamagnetic nanoparticles made of coordination networks," she said. The particles should be useful to study the limit for observing quantum tunneling of magnetization on a single nanoparticle, a key property needed for materials to function as quantum bits for computing.
The University of Paris South group, in collaboration with Corine Mathonière of France's University of Bordeaux and others, extended the processing methods to make Cu2[Mo(CN)8] nanorods. The latter compound is an example of a photomagnet, one in which magnetism can be controlled by light.
The poly(vinylpyrrolidone)-coated nanorods are paramagnetic under ambient conditions, she said, but irradiation with 410-nm light induces electron transfer from a Mo(IV) atom to one of the Cu(II) atoms, creating Mo–CN–Cu ferromagnetic units between the newly formed Mo(V) atoms and the remaining Cu(II) atoms (Chem. Commun. 2005, 746). Compared to the bulk compound, the nanoparticles undergo a complete transformation of Mo(IV) under irradiation, she added. The magnetization is reversible by discontinuing irradiation and heating the sample. Mallah and Mathonière each presented their own lectures in San Diego on related topics.

"One of the appealing aspects of cyanide-based chemistry is the ability to tailor the magnetic, electronic, and other properties, and, to a great extent, control the resulting architecture," Dunbar said. Her group at Texas A&M and Long's group at UC Berkeley have been using these advantages to create new varieties of single-molecule magnets. Most single-molecule magnets to date are manganese oxide clusters, Dunbar said, but cyanide ligands typically coordinate to two metal atoms in a linear fashion and are easier to control than oxide ligands, which can coordinate numerous metal atoms with different M–O–M angles.
Dunbar and Long each reported on their groups' efforts to make novel molecular cubes, squares, and other shaped complexes in order to use them as building blocks for larger molecular assemblies. Dunbar's work includes some of the first compounds with clusters that contain heavier transition metals, such as Mn4Re4 (C&EN, Dec. 13, 2004, page 29). Long's group has been making clusters with M4M´4(CN)12 and M6M´8(CN)24 cores that contain as many metal centers as possible (Acc. Chem. Res. 2005, 38, 325).
THE GOAL is to see which combinations of ligands and metals might sufficiently increase the magnetic anisotropy (directionality) associated with the ground state of high-spin clusters, Long explained. The level of anisotropy is directly proportional to the spin-reversal barrier, an energy barrier for conversion of magnetization from "spin up" to "spin down" states, he noted. Controlling this directionality in single molecules could enable higher density storage of information. Molecules with higher spin-reversal barriers should be capable of storing information at more practical temperatures--above the current limits of about 4 K for single-molecule magnets, he noted.
Dunbar's and Long's groups are starting to meet with some success. For example, one of Long's compounds incorporating [Mo(CN)7]4– appears to have a spin-reversal barrier higher than the record barrier of the prototypical single-molecule magnet Mn12O12 cluster, he said.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter