Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Super Sponges Soak up Co2
Custom-designed porous compounds exhibit unprecedented gas uptake
by Mitch Jacoby
December 12, 2005
| A version of this story appeared in
Volume 83, Issue 50

Materials Chemistry
Imagine a supersponge capableof soaking up vast quantities of carbon dioxide in the exhaust gas of power plants or in automobile tailpipe emissions. Such a sponge might be useful in new procedures for greenhouse-gas cleanup that are more efficient and cost-effective than current methods, which include treatment with aqueous solutions.
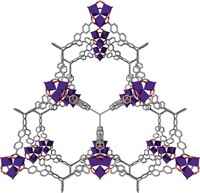
Using chemical synthesis, researchers at the University of Michigan, Ann Arbor, have created a whole family of gas-gobbling sponges. The materials, which are known as metal-organic framework (MOF) compounds, are stable, crystalline, porous substances consisting of metal clusters joined by organic linker groups. In earlier studies, the researchers characterized the crystals' structures and surface areas. Now they report that MOFs are well-suited to trapping CO2. One of the compounds, MOF-177, has a room-temperature CO2 capacity of 140 weight % (33.5 mmol per g) at moderate pressure (about 30 bar), which far exceeds the CO2 storage capacity of any other porous material (J. Am. Chem. Soc., 2005, 127, 17998).
To measure gas uptake, chemistry professor Omar M. Yaghi and graduate student Andrew R. Millward prepared samples of MOF compounds in evacuated containers and then pressurized the vessels with CO2 incrementally. At each pressure step, the gas and adsorbent were left to equilibrate and the corresponding weight change (corrected for various artifacts) was measured.
Of the large number of well-characterized MOF compounds, the Michigan group tested nine materials with widely varying structures and compositions. The list includes zinc- and copper-based crystals, compounds with square or cylindrical channels, and other samples with various-sized pores that are functionalized with amino or alkyl groups.
The supersponge, MOF-177, consists of octahedral Zn4 carboxylate clusters linked to organic groups. The material is characterized by exceptionally high surface areasome 4,500 m2 per g, which corresponds roughly to four football fields of surface area per gram of material. According to the researchers, the compound can store roughly twice as much CO2 as standard high-surface-area carbon materials under identical conditions.
Yaghi compares gas uptake in the crystals to collecting bees in a honeycomb. In the absence of a beehive, the bees swarm about freely, he explains, filling a large volume sparsely. In contrast, they pack densely in the interior of a beehive. Similarly, he points out, a cylinder filled with MOF-177 can hold nine times as much CO2 as a cylinder without the adsorbent under the same conditions of temperature and pressure.
After CO2 is trapped, the gas can be released readily through gentle heating and then used as a reagent in a variety of reactions. Some of the proposed uses include polymerization processes to manufacture polycarbonate-based construction materials and carbonation of soft drinks.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter