Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Styrene Breakthrough
Novel engineered catalyst opens up alternative route to commodity polymer feedstock
by Stephen K. Ritter
March 19, 2007
| A version of this story appeared in
Volume 85, Issue 12
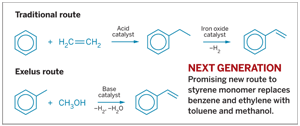
IT'S NOT EVERY DAY in the chemical industry that a potential new process for making a commodity chemical comes along and is called a breakthrough. But that's what some chemists are saying about a recently unveiled method for producing styrene monomer.
Researchers at Exelus Inc., in Livingston, N.J., have demonstrated a lab-scale process to make styrene from toluene and methanol in a single step. The route, when implemented on a production scale, could result in "dramatic reductions" in feedstock and energy costs compared with the traditional two-step route that employs benzene and ethylene as the reactants, says chemical engineer Mitrajit Mukherjee, the company's founder and president.
Styrene is an important monomer with a global demand of more than 25 million metric tons per year, Mukherjee notes. It is used to make plastics and synthetic rubbers for numerous applications.
Dow Chemical, in the U.S., and I. G. Farben, in Germany, introduced the two-step styrene process some 70 years ago, he says. In the first step, benzene and ethylene react via a solid-acid catalyst to form ethylbenzene. In the subsequent step, the ethylbenzene, mixed with high-temperature steam, passes over an iron oxide catalyst at 625 oC. The catalyst dehydrogenates the ethyl group to yield styrene.
Styrene also is commercially available as a by-product of industrial propylene oxide production from ethylbenzene and propylene.
Industrial chemists have been eager for some time to find an alternative route to styrene to solve a few unsavory problems. For one thing, benzene from crude oil and ethylene from natural gas are high-priced starting materials for producing a staple like styrene monomer. In addition, the high-temperature dehydrogenation is an energy-intensive process, which is a curse for chemical companies these days because of high energy prices. The opportunity to use less toxic toluene in place of benzene, a known carcinogen, is another plus.
A number of researchers have attempted to synthesize styrene via the toluene-methanol route during the past 30 years, Mukherjee says. But up to now, no one has been able to devise a catalyst with the type of yield and selectivity needed for a commercially viable process. Decomposition of methanol to hydrogen and carbon monoxide during the reaction was one limitation. Another shortcoming was that the hydrogen could react with styrene to re-form ethylbenzene.
According to Mukherjee, the key to the Exelus process, called ExSyM, is a novel engineered catalyst that facilitates styrene synthesis in a single step at 425 oC and atmospheric pressure. The catalyst is a proprietary zeolitic material containing basic active sites in a highly optimized pore structure that can be used in a standard fixed-bed reactor, he explains. The active sites selectively adsorb toluene over methanol to limit methanol decomposition, and the pore structure facilitates the diffusion and residence time of the reactants to enhance toluene alkylation, he says.
Styrene and ethylbenzene are produced in a 9:1 mixture in the lab-scale process, with nearly complete conversion of toluene and methanol. At production scale, the ethylbenzene by-product could be separated and sold to a conventional styrene producer, or it could be dehydrogenated on-site to boost the overall styrene yield. Hydrogen and water also are by-products. The hydrogen could be burned to produce steam to help heat the product distillation columns.
Mukherjee predicts that the ExSyM process, when implemented at full scale, could reduce styrene production costs by $350 or more per metric ton. Most of the savings would accrue from replacing benzene and ethylene with toluene and methanol, subject to feedstock cost fluctuations, he says. Additional savings of about $20 per metric ton would come from the lower energy bills made possible by the lower reaction temperature. An added environmental benefit for the overall process is significantly lower emissions of greenhouse gases, namely methane and carbon dioxide, he notes.
"Coupling the alkylation and dehydrogenation in one step is very slick," comments Leo E. Manzer, a retired DuPont research fellow who currently is president of consulting firm Catalytic Insights, in Wilmington, Del. "With no apparent catalyst deactivation and a high yield using toluene and methanol, Exelus appears to be well on the way to a breakthrough styrene process." The potential for using biomass-derived methanol to include a renewable carbon in the styrene could be another possible environmental advantage, he adds.
CHEMISTRY PROFESSOR Avelino Corma of Polytechnic University of Valencia, in Spain, likewise is impressed. In 2002, Corma and his colleagues published the results of an exhaustive combinatorial study to screen basic zeolite catalysts for toluene alkylation. The best catalysts identified by their study resulted in styrene yields of less than 5%. The researchers concluded that the toluene-methanol route might never be capable of industrially important yields.
Asked to comment on the Exelus process, Corma says it "may finally open a new route for industrial production of styrene in one step." He cautions that long-term stability of the catalyst will need to be proved first. Exelus currently is in the process of doing that by way of 100-hour stability tests.
As a technology development firm, Exelus doesn't plan to produce styrene, but it will license the technology to current styrene producers, notes James Nehlsen, an Exelus senior process development engineer. "The ExSyM process is designed to closely resemble that of a conventional styrene monomer plant, so it will be amenable to retrofitting into existing styrene monomer units," Nehlsen says. Several companies already have shown interest on the basis of recent and planned presentations at conferences, he says.
The development time for a chemical process like ExSyM is about five years, Nehlsen continues. "But the compelling economics of this particular process are accelerating that rate. We expect to begin pilot-scale work this summer."
Exelus was founded to develop and commercialize greener and less energy-intensive processes to produce commodity chemicals and transportation fuels. The "cleaner by design" research leading to the ExSyM process is being funded in part by grants from the Department of Energy (DOE) and the New Jersey Commission on Science & Technology.
Back in December, Exelus announced successful pilot-plant testing of its first technology, called ExSact, a fixed-bed reactor system containing a solid-acid alkylation catalyst. ExSact is designed to convert light hydrocarbons into alkylate, a high-octane gasoline ingredient rich in isooctane. Alkylate typically is made by using difficult-to-handle liquid sulfuric acid or hydrofluoric acid catalysts. So far, ExSact is being used to revamp a refinery unit in Europe.
Charles G. Russomanno, Small Business Innovation Research portfolio manager for DOE's Office of Energy Efficiency & Renewable Energy, calls the Exelus styrene technology "a win-win situation for styrene producers. It's a rare case of cutting carbon dioxide emissions while dramatically reducing operating costs."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter