Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Ascent Of Quadruplexes
Nucleic acid structures become promising drug targets
by Stu Borman
May 28, 2007
| A version of this story appeared in
Volume 85, Issue 22
IN 1962, when David R. Davies was in his 30s, he and two coworkers at the National Institutes of Health proposed that short, guanine-rich stretches of DNA could assume unusual structures. On the basis of diffraction studies of polyguanylic acid gels, they concluded that if four guanines were close enough together they could form planar hydrogen-bonded arrangements now called guanine quartets (G-quartets).

"One would expect this to be a particularly stable structure," they wrote at that time in the first paper on the topic (Proc. Natl. Acad. Sci. USA 1962, 48, 2013). "The existence in solution of planar tetramers of this kind could then result in the formation of linear aggregates formed by stacking the tetramers on top of each other, since the large planar surfaces would result in strong van der Waals attractions. Such aggregates"—now called G-quadruplexes—"would be roughly cylindrical in appearance."
Davies, 80, now chief of the section on molecular structure at the National Institute of Diabetes & Digestive & Kidney Diseases, never followed up on his G-quadruplex idea. But others have, and Davies has become, rather unexpectedly, the founding father of a vibrant field.
That's why Davies was invited to give the opening address at the First International Meeting on Quadruplex DNA, held last month in Louisville, Ky. The meeting was organized by biophysics professor J. Brad Chaires of the University of Louisville and professor of pharmaceutical sciences Laurence H. Hurley of the University of Arizona, in Tucson. The meeting showed "that quadruplex DNA displays a wonderful diversity of structures and that these structures seem to contribute to diverse biological functions," Chaires said.
Attendees described the latest research on topics such as quadruplex structure and modeling, the functional roles quadruplexes may play in human telomeres and promoter regions of genes, and targeting quadruplexes with therapeutic agents. Telomeres protect the ends of chromosomes, and promoters are chromosome sequences that control gene expression.
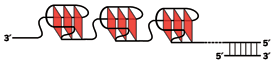
As Davies predicted, guanine-rich nucleic acid sequences are now known to be capable of adopting G-quadruplex conformations. These are roughly cubical structures that contain flat G-quartets of hydrogen-bonded guanines, similar to the tetramers Davies and coworkers proposed in 1962. If a G-quadruplex corresponds to a molecular building, then G-quartets are its floors and the nucleic acid phosphodiester backbone forms its supports.
Now researchers suspect that hundreds of thousands of DNA sequences sprinkled throughout the human genome are potential quadruplex-forming sites. And they say directing drugs to these sites might be a way of artificially regulating gene expression and thus providing medicinal benefits such as anticancer activity.
THE ANTICANCER potential of quadruplexes may arise in part from the formation of DNA quadruplexes in telomeres, where such structures have been shown to inhibit the activity of telomerase, an enzyme expressed selectively in cancer cells. Telomerase catalyzes the extension of telomeres by the addition of specific nucleotide motifs, a process that enables cancer cells to proliferate indefinitely.
Telomerase can't extend quadruplex-containing telomeres, because the enzyme doesn't recognize them as a substrate. It only copies linear strands of DNA. The resulting loss of telomerase activity causes cancer cells to lose the ability to divide, which leads to cell death.
In addition to inhibiting telomerase, quadruplexes may have a range of other important biological functions. For example, quadruplex structures may play an important causative role in Fragile X syndrome, the most common form of inherited mental retardation. DNA quadruplexes also have been implicated in the cause of two premature-aging conditions, Bloom and Werner syndromes. And quadruplexes may be present in thousands of gene promoters and thus may affect gene expression, suggesting they could exert broad influence over a wide range of processes in the body.
Most research on quadruplexes, though, has focused on their role in telomeres. In eukaryotic organisms, telomeres have single-stranded regions that include multiple TTAGGG repeat sequences (where T is thymine, A is adenine, and G is guanine), and this is exactly the kind of G-rich sequence in which quadruplexes appear.
To better understand the biological function of G-quadruplexes and guide the design of drugs that interact with them, scientists have been characterizing quadruplexes with X-ray crystallography and nuclear magnetic resonance spectroscopy (NMR). Structural biologist Dinshaw J. Patel of Memorial Sloan-Kettering Cancer Center and coworkers determined the NMR structure of a human telomeric quadruplex in sodium ion solution in 1993, for example.
When X-ray structures of human quadruplexes in a potassium-containing crystal were determined nearly a decade later by professor of chemical biology Stephen Neidle of the University of London School of Pharmacy and coworkers, they looked a lot different.

In fact, it seems that quadruplexes may adopt a variety of structures. In the last couple of years, three groups, working independently, described the folding patterns or the full three-dimensional structures of several human telomeric sequences in K+ solution. In each case, the quadruplexes' shapes were distinctly different from each other and from those found previously. This work was carried out by chemistry professor Hiroshi Sugiyama and coworkers at Kyoto University, in Japan; assistant professor of pharmacology and toxicology Danzhou Yang of the University of Arizona and coworkers; and Patel, biophysicist Anh Tu??n Phan of Nanyang Technological University, in Singapore, and coworkers.
Scientists have also studied the 3-D structures of several quadruplexes found in gene promoters. In these settings, too, the quadruplexes take on different shapes. And there may be other types of quadruplex structures floating around in cells, in addition to the ones analyzed so far, said pharmaceutical chemist Mateus Webba da Silva of the University of Ulster, in Coleraine, U.K.
In addition to cataloging the various structures of quadruplexes, it's important to know what they are doing when they move around in living cells. That's why quadruplex researcher Shozeb Haider and Neidle recently used principal component analysis and molecular dynamics simulations to create computational models of quadruplexes in motion. The simulations reveal that connecting loop sequences in quadruplexes are major contributors to quadruplex flexibility and potential sites for drug binding. "People used to ignore loops, but now they have become very interesting, and we're designing drugs based on them," Haider said.
Bioinformatics analyses of G-rich gene sequences likely to adopt quadruplex conformations suggest that quadruplexes may be much more prevalent in genomes than was formerly recognized. Using a simple folding rule for possible quadruplex structures, research fellow Julian Huppert and chemical biologist Shankar Balasubramanian of the University of Cambridge reported that human genes contain as many as 376,000 quadruplex-forming sequences. That means 43% of human genes could have a quadruplex motif in their promoter regions, which would make this "one of the most prevalent regulatory motifs known," Huppert said.
In their studies of yeast genes, F. Brad Johnson, assistant professor of pathology and laboratory medicine at the University of Pennsylvania, and coworkers also found potential quadruplex-forming sequences to be common near promoter sites. And in the most wide-ranging analysis carried out yet, Shantanu Chowdhury of the Institute of Genomics & Integrative Biology, in Delhi, India, and coworkers have found putative quadruplex-forming sequences to be plentiful in or near promoters in mouse, rat, chimpanzee, and human genes. Chowdhury cautioned that this could mean quadruplex-targeted drugs would have substantial side effects because they could affect gene regulation in a variety of unpredictable ways.
Although RNA quadruplexes have been less commonly studied than DNA quadruplexes, these RNA structures may also have important functional and clinical significance. For example, mutations in a gene called FMR1 cause Fragile X syndrome. Biochemist Michael Fry of Technion-Israel Institute of Technology, in Haifa, and coworkers have found that mRNA transcripts of FMR1 produced in Fragile X carriers can form quadruplexes that prevent the mRNAs from being translated into proteins. Marking a step toward potential treatment of the condition, Fry's group also has shown that proteins of the heterogeneous nuclear ribonucleoprotein family can destabilize these quadruplexes and enhance translation in vivo.
Balasubramanian and coworkers recently reported that an RNA quadruplex in the transcript of a human oncogene inhibits expression of that gene as well. "Given the emerging interest in small molecules that target and stabilize nucleic acid quadruplexes with specificity, this RNA G-quadruplex has obvious potential as a molecular target for small-molecule therapeutic agents," the researchers noted (Nat. Chem. Biol. 2007, 3, 218).
Indeed, a major focus of the meeting in Louisville was on quadruplex-related drug candidates. This year marks the 10th anniversary of a landmark finding by Neidle, Hurley, and coworkers that a 2,6-diamidoanthraquinone derivative could target a quadruplex structure and inhibit telomerase (J. Med. Chem. 1997, 40, 2113). This study captured the imagination of investigators and sparked interest in quadruplex-targeted agents that continues to this day.
Kazuo Shin-ya and Haruo Seto of the Institute of Molecular & Cellular Biosciences at the University of Tokyo and coworkers later isolated a quadruplex-binding bacterial natural product called telomestatin that inhibits telomerase potently and selectively (J. Am. Chem. Soc. 2001, 123, 1262). Neither 2,6-diamidoanthraquinones nor telomestatin has been developed further because they lack some key properties that characterize practical drug candidates.
Several years ago, Hurley and coworkers found the first evidence that a quadruplex structure forms in the promoter of a human gene-namely c-Myc, a gene that's actively expressed in a variety of tumor types (Proc. Natl. Acad. Sci. USA 2002, 99, 11593). This helped confirm an earlier prediction by associate professor of biomedicine Tomas Simonsson of G??teborg University, in Sweden, and coworkers that a G-quadruplex may control c-Myc gene expression. Hurley's group also showed that a small organic molecule-the cationic porphyrin tetra(N-methyl-4-pyridyl)porphine (TMPyP4)-interacts with the c-Myc quadruplex and inhibits expression of the gene.
Hurley and coworkers' c-Myc inhibition findings were recently confirmed by pharmaceutical scientists Zhi-Shu Huang and Lian-Quan Gu of Sun Yat-sen University, in Guangzhou, China, and coworkers. They showed that quindoline derivatives could induce or stabilize G-quadruplexes in c-Myc and thus inhibit the gene's expression in cancer cells (J. Med. Chem. 2007, 50, 1465).
As is the case for the 2,6-diamidoanthraquinones and telomestatin, TMPyP4 isn't druglike. And the quindoline derivatives identified by the China group are "a long way from being compounds you could take to the clinic, because they're too toxic," Hurley said.

NEVERTHELESS, his group's work on c-Myc did lead to a drug candidate, a compound called CX-3543. Researchers at Cylene Pharmaceuticals, in San Diego, designed CX-3543 starting with fluoroquinolone telomerase inhibitors discovered in Hurley's lab. The compound interacts selectively with the type of G-quadruplex found in the c-Myc promoter.
Jeffrey P. Whitten, Cylene vice president of research, noted that company scientists designed CX-3543 to bind to c-Myc and induce cell suicide in tumor cells that overexpressed the gene. Although the agent does indeed target a quadruplex, it turns out to have a novel and completely unexpected mechanism of action. It disrupts a complex of quadruplex DNA with the protein nucleolin that forms in cancer cell nucleoli. This activity, in turn, interferes with biosynthesis of ribosomal RNA in cancer cells, leading to death of the cells.
Cylene recently completed a Phase I clinical trial of CX-3543. The results indicate that the agent is safe for human use, and it will soon enter Phase II trials.
"It's great to have a drug get this far that has a novel mechanism of action," Hurley said. "Sure, I would love it if it worked exactly as we originally said"—by inhibiting c-Myc expression. "But I was extremely relieved to find that it does still work through a quadruplex," he said.

The other quadruplex-related agent in clinical trials is AS1411, a 26-base G-rich DNA oligonucleotide that folds into a quadruplex and acts as an aptamer (a nucleic acid oligomer that binds a molecular target). It was discovered serendipitously by researchers Donald M. Miller, Paula J. Bates, and John O. Trent of the University of Louisville in a study of triple-helix-binding oligonucleotides.
Advertisement
The scientists have yet to uncover the mechanism of AS1411, so they don't know whether it adopts its quadruplex form in cells. But in a tantalizing coincidence, AS1411's mechanism, like CX-3543's, involves nucleolin. AS1411 binds to nucleolin on tumor cell surfaces, apparently causing the cells to die. Normal cells lack nucleolin surface receptors and don't take up AS1411, Bates said.
In animal trials, AS1411 inhibited cancer cell proliferation and tumor growth. Phase I trials of the drug in cancer patients have so far revealed no serious toxicities, Miller said. Of renal cell carcinoma patients who took the drug, one continues to experience complete remission of nearly three years' duration, one had a near-complete remission, five showed little change, and two continue to get worse.
The Phase I trial was initiated by Aptamera, a Louisville biotech firm founded by Miller, Bates, and Trent. London-based Antisoma acquired Aptamera in 2005 and continued the trial. Antisoma will soon begin Phase II trials of AS1411 in patients with renal cell cancer and acute myelogenous leukemia.
Efforts to develop other types of quadruplex-targeted drugs continue. For example, Neidle and coworkers have been investigating the BRACO-19 family of acridine compounds, which target telomeric quadruplexes. Some of these have shown anticancer activity and have reached the preclinical development stage.
IN ADDITION, Phan, Neidle, Patel, and coworkers recently reported the NMR structure of a quadruplex that forms in c-Kit, a gene involved in gastrointestinal tumors (J. Am. Chem. Soc. 2007, 129, 4386). These tumors are currently treated with Gleevec, but patients develop resistance to the drug, Neidle said. The researchers believe that down-regulating c-Kit expression by targeting its quadruplex might help such patients. "The c-Kit quadruplex sequence is unique in the human genome and appears to form a unique tertiary structure, making it a very 'drugable' target," Neidle said.
At the meeting in Louisville, Neidle pointed out that quadruplex research has rapidly moved from fundamental science in laboratories to biomedical investigations involving human clinical trials. "It is hard to think of another area in which so much has been achieved with a new drug concept" in recent years, Neidle said.
"Much still needs to be learned about the biology of both telomeric and genomic quadruplexes, and more robust experimental data on their roles in cells are needed," he noted. "The meeting also highlighted the need for more structural studies, both to explore the wide diversity in topology shown by quadruplexes and to guide structure-based design in the future. There was a real sense that quadruplex-targeted drugs are going to be increasingly important."
Conference coorganizer Chaires added that he thought the meeting "provided an accurate snapshot of where research on quadruplex DNA now stands. Quadruplex DNA is no longer just a biophysical oddity and must be given serious consideration as a valid target for new types of drugs."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter