Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Brain Barricade
Researchers are tackling the grueling challenge of getting brain therapies across the blood-brain barrier
by Sarah Everts
June 4, 2007
| A version of this story appeared in
Volume 85, Issue 23

GIVEN ITS COLOSSAL TASK of masterminding the body's every move, one really can't blame the brain for being strict about chemical security.
Elsewhere in the body, nutrients and drugs diffuse freely between the bloodstream and surrounding tissues, often maneuvering through the spaces between cells. Not so between the blood and the brain. Compared with many other tissues, our gray matter exists in a chemical sanctuary set apart from the blood: What gets in and what gets out is tightly regulated, with good reason.
Neurons must be protected from potential toxic compounds in the bloodstream and from the chemical fluctuations that occur after a meal or exercise that might disrupt the ionic niche essential for optimal performance.
At the same time, the brain needs a constant supply of glucose, oxygen, and other nutrients to feed these energy-intensive neurons. In fact, our gray matter boasts an extraordinarily extensive circulatory system: A given neuron is never farther than 20 µm away from a capillary.
To balance the need for nutrients and a safe niche, the mind relies on a stalwart chemical checkpoint called the blood-brain barrier (BBB). Despite its monolithic-sounding name, the BBB is simply a tremendously tight-knit layer of endothelial cells.
These cells coat the 400 miles of capillaries and blood vessels in the brain, creating a barricade that is four times longer than the former Berlin Wall. The nearly impermeable junctions between BBB cells are formed by the interdigitation of about 20 different types of proteins, creating "the highest resistance barrier in biology," says William Pardridge, director of the Blood-Brain Barrier Research Laboratory at the University of California, Los Angeles, School of Medicine. "The best analogy is that of a zipper," he says. "Neighboring cells of the BBB have protein teeth that lock together like a zipper."

Molecules must enter a BBB cell through membrane-embedded protein transporters or by slipping directly through its waxy outer membrane. Once inside, foreign compounds must avoid a high concentration of metabolic enzymes and a variety of promiscuous protein pumps primed to evict any riffraff. Having avoided these obstacles, foreign molecules must then pass through the inner membrane of a BBB cell to finally reach the brain.
These elaborate defenses allow the BBB to sequester the brain from potential harm. But the BBB also obstructs delivery of neurological drugs to a site of disease in the brain.
"The BBB poses a major challenge for many drug development programs," says Edward Kerns, associate director of chemical and screening science at Wyeth Research. "The same features of a molecule that might make it active for a particular therapeutic target sometimes will restrict it from getting into the brain."
"Whenever someone is developing a drug for the brain, at some point, they'll have to bang up against the BBB," says Quentin R. Smith, School of Pharmacy chair at Texas Tech University Health Sciences Center. By some estimates, more than 90% of potential drug candidates are excluded from the brain by the BBB.
Still, researchers in academia and the biotech and pharmaceutical industries are learning to bypass the BBB or fool it into letting potential drugs into the brain. They are designing small drugs that can passively diffuse through the BBB or piggyback on nutrient transporters to get inside the brain. Others are attaching potential therapeutics to Trojan horses that the brain will unwittingly engulf.
Most Food & Drug Administration-approved brain pharmaceuticals, such as antidepressants and antinausea agents, get into the brain by passive diffusion across the BBB's inner and outer cell membranes. Nicotine, alcohol, cocaine, heroin, and most other recreational drugs get in the same way. Molecules that slip across the membrane of BBB cells are typically small (under 500 daltons), are lipid-soluble, and have a low polar surface area. They also tend to have few potential hydrogen-bond-forming moieties, including few, if any, carboxylic acid groups.
Tweaking a moiety or two can have dramatic results. Heroin is a case in point. It penetrates the BBB 100 times more effectively than does morphine, making it far more addictive than morphine. The only chemical difference between these molecules is that morphine has two hydroxyl groups, whereas heroin has acetyl groups instead. This acetylation makes heroin much more lipid-soluble, allowing it to readily diffuse across the BBB.

"Designing a good brain drug requires balancing many physicochemical and metabolic properties at once," Kerns says. For example, one might be tempted to focus just on making a drug lead extremely hydrophobic, as was the early strategy for many medicinal chemists working on delivery to the brain.
Hydrophobic molecules tend to make nonspecific interactions with a variety of proteins and lipids and thus can divert a potential drug away from its intended target, says Stephen Hitchcock, director of medicinal chemistry at Amgen. "Just getting into the brain may not be enough." Ideally, a drug reaches its target without making "a lot of nonspecific interactions," he says.
Even so, a drug's potency can make up for the nonspecific interactions it makes. Consider antidepressants. Both fluoxetine (Prozac) and sertraline hydrochloride (Zoloft) cross the BBB "like gangbusters," says Hitchcock, but they also make a lot of nonspecific interactions. The antidepressants still achieve a pharmacological end because the small fractions that aren't diverted by nonspecific interactions are enough to potently inhibit serotonin uptake. Potency likewise compensates for pitiful BBB penetration. For example, only a small fraction of morphine crosses the BBB, yet its potency allows it to remain a powerful narcotic.
Now consider this nightmare. Everything could be seemingly perfect—your drug passively diffuses across the BBB cell membrane, it doesn't interact randomly with other proteins, and it's extremely potent. "Then suddenly the 800-lb gorilla appears," Smith says. He is referring to P-glycoprotein (PGP), the most notorious among a suite of BBB efflux transporters that counter passive diffusion by pumping foreign entities out of the brain. PGP has been shown to mediate expulsion from the brain of more than 30 drugs, including antitumor agents and antibiotics.
"Evading the efflux transporters is a tremendous challenge for medicinal chemists," Hitchcock says. "P-glycoprotein has evolved to recognize a huge diversity of substrates. It is very promiscuous. Once you have a compound that's a PGP substrate, it is very difficult to engineer out PGP recognition."
Medicinal chemists have come up with some empirical rules to avoid PGP expulsion. One such rule is that the sum of nitrogens and oxygens in the structure should be eight or fewer. But these rules are not hard and fast; they are more like rules of thumb.
And although developers of brain-targeted drugs may curse these efflux transporters, colleagues developing therapeutics for other organs count their lucky stars when their pharmaceutical leads are pumped out of the brain by PGP, thereby preventing neural side effects.
In fact, the sleep-inducing difference between sedating and nonsedating antihistamines is tied to BBB efflux proteins. Pfizer scientists have shown that both kinds of antihistamines passively diffuse across the BBB. But sedating antihistamines are not expelled by PGP and elicit their sedating side effects by staying in the brain.
On the other hand, second-generation, nonsedating antihistamines are serendipitously exported by PGP. In one case, the difference between PGP recognition of a sedating antihistamine compared with a nonsedating counterpart is due to conversion of a CH2OH group in the former into a carboxylic acid in the latter.

ALTHOUGH PASSIVE diffusion remains the preferred pharma route into the brain, there are a few FDA-approved small-molecule drugs that piggyback on protein transporters to cross the BBB. The drugs L-Dopa, to treat Parkinson's disease, and gabapentin, to treat epilepsy and pain, both hijack an amino acid transporter to cross the BBB barricade, a mechanism that developers only realized was in play after the fact.
Besides importing amino acids, other BBB membrane proteins specifically transport nucleosides, sugars, and choline from the blood into the brain. Physicians co-opt a hexose transporter that usually hauls glucose into the brain to instead deliver fluorodeoxyglucose and other sugars used for positron emission tomography (PET) scanning of the brain. Some researchers have conjugated sugars to peptide drugs to encourage uptake by this transporter. Biotech companies such as Santa Clara, Calif.-based XenoPort are also evaluating nutrient transporters for use in brain drug delivery.
One challenge in exploiting such transporters to get small- or smallish-molecule drugs into the brain is that chemists have to optimize several pharmacophores simultaneously, Hitchcock points out. "You are optimizing for recognition of the transporter but you also have to preserve biological activity at the target," all the while steering clear of efflux proteins, he notes.
Currently, the driving force in the pharmaceutical industry for brain therapeutics is small drugs, and "it will be like this for quite a while," Texas Tech's Smith predicts. "But in the long term, biologics—including antibodies, proteins, enzymes, nanoparticles, and gene therapy—will be the drugs of choice."
Pardridge argues that big pharma should start planning for brain delivery of these larger sized therapeutics now. If large molecules are ever to be targeted to the brain, Pardridge notes, developers of brain drugs need to seriously consider alternative ways of crossing the BBB besides passive diffusion and the mediated protein transport that works for small molecules.
As such, Pardridge and others are investigating, and trying to exploit, the way large molecules are smuggled across the BBB. In particular, they are looking at a mechanism called receptor-mediated endocytosis, which imports insulin, peptides, and iron-carrying proteins from blood into the brain.
In receptor-mediated endocytosis, receptors on the membrane of a BBB cell detect the presence of a compound normally ushered into the brain. This initiates engulfing of the receptor and its ligand into a vesicle that forms at the surface of the BBB cell and then enters the cell.
Because this form of import does not depend on the size or shape of what is being engulfed, this route across the BBB provides a promising strategy for delivering bulky drug payloads, says Pardridge, whose lab pioneered what is now known as the Trojan horse technique of crossing the BBB.
Partridge's method relies on an antibody engineered to recognize a BBB receptor for insulin. Antibody binding to the receptor activates endocytosis. So far, his group has successfully sent 100-kDa enzymes and liposomes filled with plasmid DNA across the BBB of animal models by conjugating these large payloads to the insulin antibody, the "horse" in this case, and injecting the macromolecular subterfuge into the bloodstream with a needle.
ArmaGen Technologies, a spin-off company from Pardridge's lab, is developing the technique to get a stroke drug into the brain. The Santa Monica, Calif.-based company hopes that clinical trials will begin in 2008. Company scientists have also used the insulin-receptor Trojan horse to deliver antibodies against the peptides that cause Alzheimer's plaques into the brains of animal models.
In addition to ArmaGen, a handful of other biotech companies have sprung up to capitalize on endocytosis-mediated delivery. For example, to-BBB, a Leiden, Netherlands-based start-up, is focused on delivering liposomes full of a Japanese encephalitis treatment to the brain via the Trojan horse technique. The treatment has been shown to kill the virus, but when doctors inject it into patients, it doesn't cross the BBB in time to be curative.
To-BBB's strategy is to get the antiviral in more quickly by inducing endocytosis in the same way as the brain-penetrating bacterial toxin that causes diphtheria does, says Alfred N. Nijkerk, chief business officer of to-BBB. Diphtheria toxin binds to a membrane protein called EGF-like growth factor and in doing so induces endocytosis. To-BBB conjugates a nontoxic mimic of this toxin to a liposome full of the antiviral. They hope it will serve as a proof of principle for their delivery approach.
Earlier this year, Brian J. Spencer and Inder M. Verma of the Salk Institute for Biological Sciences took the Trojan horse strategy a step further. They were trying to use the technique to get an enzyme that cures a lysosomal storage disorder across the BBB, but they wanted to sidestep the need for regular intravenous injections of the enzyme.
Advertisement
So they decided to design a drug that would get a patient's liver to produce the therapeutic itself. To do so, they connected the gene for the therapeutic enzyme with a gene for a peptide sequence that initiates endocytosis at the BBB. The two-gene chimera was put into a virus and injected into animal models. The genes were inserted into liver cells, which began to produce the Trojan horse treatment. The therapeutic agent even managed to cross the BBB, albeit in modest amounts.
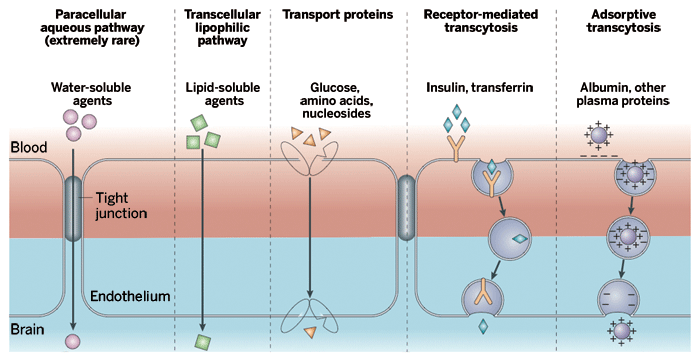
ITS PROMISE notwithstanding, there are several challenges to the endocytosis delivery method. One is the high cost. Another is that it remains difficult to target the drug specifically to BBB endothelial cells. For example, insulin receptors are not unique to the BBB cells, and precious treatment could be wasted by delivering payloads to non-BBB cells. Another concern is that sending drugs across the BBB through receptor-mediated endocytosis might compete with, or at the very least perturb, transport of essential nutrients into the brain.
Many researchers point to BBB proteomics as an important step toward increasing the portfolio of available strategies for crossing the BBB. Proteomic studies could facilitate the identification of BBB-specific receptors, which would permit drugs that are too expensive to be administered systemically to be targeted solely to brain tissue, says Joan Abbott, a BBB researcher at King's College London. Furthermore, she argues, identifying all the transport and efflux proteins in the BBB cell membrane would provide drug developers with a more complete understanding of their friends and foes.
The challenge is that proteomics of the BBB involves primarily membrane proteins, which are not easy to analyze with standard proteomics techniques such as two-dimensional gel electrophoresis, says Eric V. Shusta, a biological engineer at the University of Wisconsin, Madison. "New tools to do BBB proteomics are essential to do it at all."
Shusta recently reported a method for engineering a troupe of antibodies that target membrane proteins found in the BBB. Using this technique, Shusta found a previously unknown BBB receptor that he is now evaluating as a new Trojan horse target.
But even as researchers discover and investigate new BBB secrets and crossing strategies, there remains a dearth of brain disease treatments that can get across the BBB. In some cases, this has driven doctors to try drastic measures to deliver brain chemotherapies, including using hyperosmotic solutions to make the BBB more permeable or blocking efflux transporters. The problem of delivering brain therapeutics will only get worse as more people develop age-related brain diseases such as Alzheimer's disease, for which there is currently no cure.
Pardridge argues that the realization of BBB-crossing therapies will require far more researchers focused on teasing out the secrets of this all-important barrier. Others in the field acknowledge the difficulty of the task but remain optimistic.
"Conditions of neurodegeneration are not well-served by current therapies, and they place a tremendous burden on our society," Hitchcock says. "Crossing the BBB is certainly not impossible, but it remains a significant challenge."
- Brain Barricade
- Researchers are tackling the grueling challenge of getting brain therapies across the blood-brain barrier
- WEB EXCLUSIVE: Appraising Penetration
- Drug Developers Rely On A Variety Of Tools To Figure Out Whether Their Lead Gets Into The Brain And Then To Its Target




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter