Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Exotic Molecule
Dipositronium molecule contains only electrons and positrons
by Elizabeth K. Wilson
September 17, 2007
| A version of this story appeared in
Volume 85, Issue 38
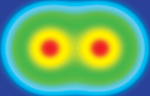
WHEN AN ELECTRON meets its positively charged antimatter counterpart, the positron, they've been known to briefly form an exotic "atom" known as positronium before annihilating each other within nanoseconds in a burst of gamma rays.
Now comes an even more complex matter-antimatter entity: a molecule of two positronium atoms.
Positronium is somewhat analogous to hydrogen, with the positron playing the role of the proton. It even has a chemical symbol, Ps. Since the 1940s, scientists have speculated about the possibility of creating Ps2 molecules.
Physicists David B. Cassidy and Allen P. Mills Jr. at the University of California, Riverside, finally orchestrated this marriage of opposites on the internal surface of a thin silica film (Nature 2007, 449, 195).
They collected positrons generated during the beta decay of sodium-22 and shot them onto the film, where the positrons linked up with electrons in the silica pores to form positronium. The energetics of the silica surface then allowed pairs of positronium atoms to bond, albeit briefly. Ps2 exists for about a quarter of a nanosecond before the electrons and positrons destroy each other.
Haruo Saito, physics professor at the University of Tokyo, remarks that such experiments are difficult, but he believes "that molecular positronium is certainly produced in their experiment."
Cassidy and Mills' work has "profound implications" for one alluring goal: to create a gamma-ray laser, note Northwestern University physicists Arthur J. Freeman and Rolando Saniz. Physicists believe that a dense-enough soup of positronium atoms should form a Bose-Einstein condensate, and an even denser, radiation-stimulated mixture could annihilate itself in an extremely intense, coherent pulse of high-energy photons.
The research invites speculation about even more exotic matter-antimatter combinations, physics professor Clifford M. Surko of the University of California, San Diego, tells C&EN. His group, which developed some of the technology used by Cassidy and Mills, studies, among other things, the binding of positrons to molecules.
"We're beginning to ask questions about the chemistry of antimatter and the chemistry of matter-antimatter interactions," Surko says.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter