Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
A Role For C-Peptide
Peptide formed during insulin production facilitates glucose clearance and may benefit diabetic patients
by Celia Henry Arnaud
January 14, 2008
| A version of this story appeared in
Volume 86, Issue 2
A PEPTIDE FORMED as part of insulin biosynthesis may have biological activity of its own and could have therapeutic benefits for patients with type 1 diabetes, who don't make sufficient insulin.
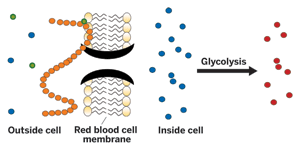
When a metal ion (green) such as Zn(II) binds to C-peptide (orange), the complex can activate the glucose transporter protein GLUT1 (black) in the red blood cell membrane. GLUT1 ushers glucose (blue) into the red blood cell, increasing glucose consumption and adenosine triphosphate (red) production in the cell.
Called C-peptide, this 31-amino-acid peptide is cleaved from the insulin precursor during synthesis of insulin, a peptide hormone that helps cells absorb glucose from the bloodstream. For a long time, researchers thought C-peptide had minimal biological activity, and they dismissed it as a by-product of insulin production. Because C-peptide is secreted at the same concentration as insulin, it has been used as a marker for endogenous insulin secretion and the health of insulin-producing pancreatic beta cells.
But C-peptide may play a larger role than as just an insulin marker. In recent years, scientists have shown that C-peptide has beneficial effects in patients with type 1 diabetes, including improved kidney function, nerve function, and blood flow. Researchers hope that administration of C-peptide might help alleviate some complications of type 1 diabetes.
Scientists don't know how C-peptide exerts its beneficial effects. Chemistry professor Dana M. Spence and colleagues at Michigan State University have found a clue to help solve that mystery. In assays where they combine C-peptide with isolated red blood cells from diabetic patients, they find that C-peptide facilitates glucose clearance and causes red blood cells to release adenosine triphosphate (ATP), a known stimulus for the blood vessel dilator nitric oxide (Diabetologia 2008, 51, 175). C-peptide causes these effects by activating the GLUT1 transporter, a protein that shuttles glucose across cell membranes.
Spence suggests that C-peptide may provide a way for glucose to enter red blood cells, which, unlike other cells, don't have insulin receptors and don't respond to insulin. If a receptor for C-peptide exists, Spence says, it may be on red blood cells but not other types of cells. He would not be surprised, however, to find that C-peptide doesn't have a receptor. No receptor for C-peptide has yet been found.
Previous work suggested that any effects of C-peptide require the presence of insulin as well, but Spence and his colleagues see the increased ATP release even in the absence of insulin. The catch is that C-peptide must first be activated by a transition-metal ion.
Spence and coworkers stumbled upon the metal connection. When they reconstituted commercial C-peptide, its activity would disappear after a day. Spence took a vial of fresh C-peptide to his colleague Gavin E. Reid, a mass spectrometrist at Michigan State, who told him that the C-peptide had Fe2+ in it. They suspect that the iron was a trace impurity from the manufacturing process.
Spence and coworkers now incubate the peptide with metal ions in water before mixing it with the physiological buffer for their assays. "The bloodstream has millimolar levels of metals that don't activate" this peptide, Spence says. Ions such as sodium and potassium could prevent the binding of metals that do activate the peptide. Spence speculates that the metal binding could explain why clinical effects of C-peptide have been so inconsistent.
John Wahren, chief scientific officer at the Swedish company Creative Peptides, has long hunted for biological functions of C-peptide. The findings suggest that when exposed to metal ions, "C-peptide may assume a secondary structure that facilitates its binding to cell membrane structures," he says. "In aqueous solution, C-peptide has an unordered structure, and metal ions may facilitate the conversion into a bioactive form."
Spence has already turned his attention to zinc as the most likely metal for C-peptide to bind to under physiological conditions. "In the beta cells, where the C-peptide and insulin are produced, the concentration of zinc is millimolar," he says. "There's a rich pool of zinc right where the C-peptide is produced."
SOME PEOPLE have been skeptical about a beneficial role for C-peptide because many patients with early-stage type 2 diabetes have elevated levels of C-peptide. Spence thinks that is because the red blood cells of diabetic patients are stiffened. "We have data showing that when the cells get stiff, the C-peptide doesn't work. If the C-peptide doesn't work and it's not facilitating glucose transport into the red cells where it is consumed through glycolysis, one may become hyperglycemic," he says. "You always hear people talking about insulin resistance, but I also think people may be C-peptide resistant."
The study is "a significant contribution to our understanding of C-peptide biology," says Anders Sima, a professor of pathology and neurology at Wayne State University, in Detroit, who studies C-peptide. The findings "may have an impact on future development of C-peptide formulations for pharmaceutical purposes."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter