Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Rubber Heals Itself
Small molecules yield self-mending property
by Rachel A. Petkewich
February 25, 2008
| A version of this story appeared in
Volume 86, Issue 8
SEVER, UNITE, STRETCH, and repeat. That's what you can do with a new class of materials called self-healing rubber (Nature 2008, 451, 977).
Chemistry professor Ludwik Leibler and colleagues at the Paris-based Industrial Physics & Chemistry Higher Educational Institution have created small-molecule-based supramolecular network materials with rubberlike properties that can repair themselves at room temperature. Rubber elasticity is traditionally a property of macromolecules, as small-molecule assemblies are generally too crystalline to bend. And although self-healing materials have been made before, they usually require heat to repair cracks and breaks.
The new small molecules—which are made from fatty acids and a combination of one, two, or three different amide groups—hydrogen-bond to one another to form the supramolecular networks. Small molecules that assemble in a network and undergo reversible cross-linking can behave like rubber, Leibler says. "The self-mending property is possible thanks to the presence of a long-lived and large excess of nonlinked, open hydrogen bonds on the fractured surfaces," he explains. The pieces must be physically placed together to mend.
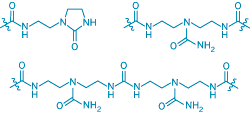
Self-healing supramolecular networks assemble from fatty acid-based small molecules containing combinations of three amides-amidoethyl imidazolidone (top left), di(amidoethyl) urea (top right), and diamidotetraethyl triurea.
Leibler says the new materials are not adhesives, because only cut or broken surfaces attach. The surfaces can self-heal if they have been apart less than one week at room temperature, or thermal equilibrium will prevent hydrogen bonding.
From a chemistry point of view, using fatty acid derivatives to build multifunctional materials and new functional groups able to associate via multiple hydrogen bonds clearly opens a new avenue for product development, Leibler says.
Polymer chemist Krzysztof Matyjaszewski of Carnegie Mellon University commends the researchers' use of a simple and renewable resource—vegetable oil—as a source of fatty acids. He says previous self-healing material systems have generally relied on complex chemistry and processing.
Arkema, a French chemical maker, is planning to develop commercial products based on the new supramolecular chemical technology.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter