Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Iron Complex Cleaves C–H and O–H Bonds
Novel compound is the first iron complex capable of activating the O–H bonds of aliphatic alcohols
by Celia Henry Arnaud
April 6, 2009
| A version of this story appeared in
Volume 87, Issue 14
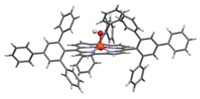
A novel diiron(IV) complex exceeds the bond-breaking capabilities of its predecessors by being able to cleave strong C–H and O–H bonds, reports a research team led by Lawrence Que Jr. of the University of Minnesota, Twin Cities, and Eckard Münck of Carnegie Mellon University (Nat. Chem., DOI: 10.1038/nchem.162). The chemists note that the complex is the first iron complex that can activate the O–H bonds of aliphatic alcohols. They obtained the complex, which has a bridging oxo group between the iron atoms, by electrochemically oxidizing a diiron(III) precursor. The diiron(IV) complex cleaves C–H bonds with bond energies as high as 100 kcal/mol. For example, under anaerobic conditions the complex oxidizes cyclohexane to cyclohexene and 1,3-cyclohexadiene. The lack of a hydroxylated product is probably due to the bridging oxo group, the researchers suggest, because forming an alcohol would require breaking both Fe–O bonds. As for cleaving O–H bonds, oxidation of tert-butyl alcohol to form acetone indicates that the diiron complex targets the hydroxyl group instead of one of the weaker C–H bonds.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter