Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Harnessing Light
Despite centuries of research, photosynthesis still has many unsolved mysteries
by Sarah Everts
April 13, 2009
| A version of this story appeared in
Volume 87, Issue 15

FROM THE SUNLIT CANOPY of dense jungle foliage to the very depths of the ocean, nature has found a way to capture the energy of light and convert it into chemical fuels.
Because of its fundamental importance, photosynthesis has for centuries challenged scientists to untangle its mysteries. Yet several Nobel Prizes for photosynthesis-related discoveries later, major unknowns still exist. Scientists have not figured out exactly how plants use light to catalyze the splitting of water, which kick-starts the entire photosynthetic process. Knowing in structural detail how Earth-abundant metals manganese and calcium can break apart water's strong bonds would be a major feat.
Water-splitting is key to the renewable production of hydrogen gas and other energy fuels, and doing so with inexpensive catalysts, as plants do a billion times per day, would be a huge step forward for solar power research. But the photosynthetic process has some other secrets, too, that scientists are only just figuring out, such as how photosynthetic organisms can tame light without suffering too much radiation damage, the plant equivalent of a sunburn.
Photosynthesis transformed Earth's atmosphere. "After the origin of life, it is the single most important transition on Earth," says Robert E. Blankenship, a plant biochemist at Washington University in St. Louis. As plants and bacteria began using the sun as an energy source, the incidental release of oxygen revolutionized our planet's atmosphere, allowing advanced life such as humans to evolve.
From cyanobacteria to plants and even in ocean corals and tiny slugs, photosynthetic organisms have tuned their light-harvesting machinery to suit "whatever wavelength or intensity of light" hits their living quarters, says Devens Gust, a photochemist at Arizona State University. Yet much of the basic photosynthetic infrastructure is standardized, he says.
To make photosynthesis possible, light must first be captured by antenna systems, or pigments. In plants, these systems sit in the membrane of organelles called chloroplasts; in bacteria, they sit on the cell membrane. Chlorophyll and its variants are by far the most common pigments in land plants, algae, and bacteria.
Chlorophyll is extremely sensitive to red and blue light, but not to green light, instead reflecting it back so that the pigment appears green to humans. Many organisms expand their light-harvesting repertoire by adding green-sensitive pigments such as carotenoids to their portfolio.
Organisms also customize pigments to match their lifestyle. For example, many marine photosynthetic organisms employ phycobilins to capture light because these pigments absorb the blue-green light that trickles through seawater. And although solar light doesn't penetrate to 1,500 meters below sea level, hot hydrothermal vents at those depths emit geothermal light at the very edge of the red spectrum. Nearby, specialized bacteria capture the handful of photons per day that emerge from the vents with self-assembling pigments called chlorosomes, Blankenship says.
The main job qualification for photosynthetic pigments is that they possess a source of free electrons that light energy can excite. When incident light energy hits a pigment, it forms an electron paired with a hole, with reductive and oxidative potential, respectively. Groups of light-harvesting pigments are attached to a mammoth, membrane-embedded 20-protein complex called photosystem II (PSII). Other pigments connect to another light-harvesting complex called photosystem I.
Photosynthesis begins when the excitation energy of each pigment is directed into the reaction center of PSII, to be used by a manganese-calcium catalyst to split water. In particular, the energy rips four protons and four electrons from two molecules of water, leaving one molecule of oxygen gas to diffuse away as a by-product. PSII drops off the protons on one side of the membrane, creating a concentration gradient that drives a molecular machine called ATP synthase. This enzyme orchestrates the production of the ATP (adenosine triphosphate) required to make sugar fuels and fulfill other energy needs of the plant.
Quinones nab the electrons and then diffuse through the membrane to another membrane-embedded protein complex. This complex deposits two more protons, which have tagged along with the electrons, on one side of the membrane, boosting the proton concentration gradient.
From there, the electrons move to photosystem I, where the light-excited electrons of the pigments are not used to split water. Instead, the energy from these electrons boosts the energy of the quinone-borne electrons. With the added punch, the excited electrons help create NADPH, an essential molecule in redox reactions to make sugar and other biological compounds.
THE ENTIRE photosynthetic process involves "more than 50 distinct chemical transformations, each mediated by at least one or two proteins," Blankenship says. When you account for helper proteins, more than 100 different proteins in total are involved, he adds.
Indeed, "a large proportion of a genome of a photosynthetic organism is devoted to photosynthesis," says Krishna K. Niyogi, a plant biochemist at the University of California, Berkeley. Yet only a fraction of those genes encode subunits of the light-harvesting photosystems, Niyogi says. Many more genes make proteins that are involved in the global assembly and regulation of the photosynthetic process, the precise roles of which "we are only just starting to figure out," he adds.
Some of these genes are responsible for maintaining a plant's circadian rhythms, which anticipate when the sun will shine and therefore ensure that adequate photosynthetic machinery is available to capture light. Others are responsible for producing the repair machinery that keeps the photosynthetic reaction centers in tip-top shape.
Repair is crucial because too much sunlight can harm the photosynthetic machinery, just as it harms human skin. "What is remarkable is that when the light energy comes in and is absorbed by the system, it is a huge blast of energy," Blankenship says. It takes an oxidant with an electric potential of at least 1 V to split water, he notes, and that's happening at multiple protein complexes at any given time. With the amount of energy pouring into the system and the possibility of creating reactive oxygen species, photosynthesis is "perhaps the most reactive chemistry happening inside any biological cell," he points out.
"The photosynthetic system has to process this energy and deal with it in an efficient way—in a sense taming it so that the energy doesn't do too much damage," Blankenship adds.
Yet some damage is inevitable. Under full sunlight, PSII needs to be repaired every 30 minutes, Niyogi explains. Researchers are only just beginning to understand how plants sense photodamage and then repair it; so unsurprisingly, these areas are subject to controversy, he adds.
What's clear is that the plant tries to avoid photodamage at the front end of the process by sensing when too much light is hitting the antenna system. It does so by measuring the pH inside the chloroplast. (Remember that one outcome of the water-splitting reaction in PSII is the transport of protons across the membrane to create a concentration gradient.) When the back-end machinery that makes ATP from this proton gradient can't keep up, too many protons build up and the pH begins to drop dangerously. The plant takes note by means of a protein sensor with a glutamate residue that gets protonated at low pH.
HOW TO SIGNAL is relayed to the light-harvesting machinery, however, "is the subject of some debate," Niyogi says, but it seems to be associated with a pigment called zeaxanthin. Some people think that zeaxanthin acts indirectly, perhaps allosterically, to shut down the harvesting of light. Others think that zeaxanthin directly disarms light-excited electrons that are on their way to the photosynthetic reaction centers.
Even as some scientists try to figure out how plants regulate photosynthesis, others hotly debate what key parts of the well-studied PSII look like.
In the early 2000s, a flurry of PSII structure determinations were achieved, first at 3.8 Å by a German team; then at 3.7 Å by a Japanese team; and finally at atomic resolution, 3.5 Å, by James Barber and his colleagues at Imperial College London in 2004.
Researchers continue to report improved-resolution structures of PSII—most recently at 2.9 Å by Wolfram Saenger's group at the Free University of Berlin and his collaborator Athina Zouni at Berlin's Technical University. But a small corner of the protein complex remains steadfastly controversial. In particular, the catalytic center that splits water is structurally unresolved, and several competing propositions for its mechanism circulate.
"Structural biologists have done heroic work on PSII, and important information has come out of the structures, but the most important part, the part we really want to see, has proven to be so difficult to resolve," says Leif Hammarström, a photochemist at Uppsala University, in Sweden.
The catalytic center splits water with the help of manganese and calcium, both of which are abundant and cheap. Its chemistry could pave a route to turning water into hydrogen and oxygen gas, which could be stored as fuels, or the redox potential of the high-energy electrons ripped off water could be harnessed in other processes to make liquid fuel such as methanol from CO2. In both cases, splitting water to produce fuels whose consumption produces water once again, "closes the loop," for humanity's energy needs, Arizona State's Gust says.

AT PRESENT, man-made water-splitting catalysts primarily rely on metals that are too expensive for market adoption and not plentiful enough to fulfill the entire world's energy demands. Figuring out how plants use cheap and abundant metals to split water would be a monumental achievement, says Gary Brudvig, a chemist at Yale University.
And everyone is focused on PSII because nature has not been flexible about water splitting. "There's no analog to the water-splitting reaction by PSII," Brudvig notes. "Oftentimes you find various versions of how nature has solved the problem. One enzyme uses one cofactor, and another enzyme uses another. That's not the case in water oxidation. Nature has one solution to the problem, solved 2.5 billion years ago. The process is absolutely conserved."
The problem with resolving nature's water-splitting catalytic center is that during X-ray bombardment of PSII crystals, the catalytic metal cluster is a magnet for any electrons in the system. Therefore, "all X-ray diffraction data suffer from inherent radiation damage, leading to reduction of Mn3+ and Mn4+ to Mn2+," cautions Saenger in the report of the most recent 2.9-Å structure, which was also solved by X-ray crystallography (Nat. Struct. Mol. Biol. 2009, 16, 334).
Other structural biologists are more convinced that the X-ray data are reliable. "We do everything we can to try to prevent radiation damage," Barber says. "We freeze the crystal to reduce damage and irradiate for a short time on a tiny part of the crystal."
Potential errors due to radiation damage notwithstanding, structural biologists have proposed several possible arrangements for the catalytic cluster. Other researchers, including Yale's Brudvig and Vittal K. Yachandra of Lawrence Berkeley National Laboratory, have used a potpourri of spectroscopic methods and modeling to try to confirm or propose the coordination environment of the metal ions in the cluster. But consensus in the field has not yet been forthcoming.
"Everybody does agree that there are four manganese atoms and one calcium atom in the catalytic site, but exactly how they are arranged is a matter of debate," Brudvig says. "It's also clear that all of the manganese ions are in high oxidation states and bridged by oxides," he says. But the exact oxidation state of each manganese atom and the precise oxide connections also remain controversial.
Barber and Brudvig favor a cuboidal core of Mn3CaO4 with a "dangler" manganese ligated to a corner oxide ion. Alternatively, Saenger and Yachandra favor a cluster with a pyramidal core, with three manganese atoms on the base, a calcium at the apex, and another manganese atom jutting off from a base manganese.
Another contentious area is the question of how efficient photosynthesis actually is—so contentious in fact that a conference is being organized this spring to resolve the issue "once and for all," says Daniel G. Nocera, a chemist at Massachusetts Institute of Technology.
Advertisement
Depending on whom you ask, "different people quote efficiencies that range from 100% to 0.1% or less," says Blankenship, who thinks the problem is mostly one of semantics. "People are talking apples and oranges because when you say 'efficiency,' it needs to be qualified." There are different types of efficiency, he points out. For example, there is quantum efficiency, which relates to how well the plant captures and processes a photon of light. Photosynthesis has extremely high quantum efficiency, near 100%, Blankenship says.
But as the excited electrons get funneled to the PSII reaction center, the efficiency of splitting water and performing the initial stages of photosynthesis is only about 30%, Gust says. When it comes to storing the energy from sunlight as sugars, plants become about 2–3% efficient.
"Have you ever seen a fat plant? That's what you do when you store energy, you become fat," Nocera says. "A plant is no different from us. Photosynthesis is very efficient, but plants don't store energy efficiently."
One reason that plants don't store fuel efficiently is that "plants' goals are different than our goals," Gust says. "The plant's goal is to live and reproduce, not to store energy for humans."
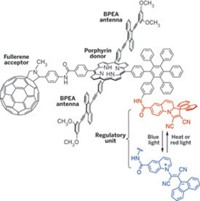
Even so, many researchers turn to photosynthesis for inspiration on how to achieve humanity's energy needs. They hope to mimic the early, energy-efficient light-harvesting steps of photosynthesis, but then direct the harnessed light energy entirely toward producing fuel instead of growing a plant.
"When we think about ways to harvest sunlight," Gust says, "it seems natural to look to photosynthesis for ideas. It has been around for the longest time, and it works on the largest scale."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter