Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Formation Of Famous Tantalum Complex Is Nailed Down
After 35 years, pentaneopentyltantalum is confirmed as the precursor to the tantalum alkylidene complex that led to Schrock metathesis catalysts
by Stephen K. Ritter
June 15, 2009
| A version of this story appeared in
Volume 87, Issue 24
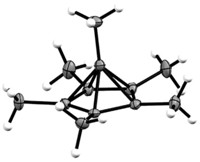
Pentaneopentyltantalum, TaR5, where R is the neopentyl group CH2C(CH3)3, may be one of the most important chemicals you have never heard of. It is now the confirmed precursor to the tantalum alkylidene R3Ta=CHC(CH3)3, the first isolated and characterized transition-metal alkylidene complex, which in 1974 launched MIT chemist Richard R. Schrock on the road to the 2005 Nobel Prize in Chemistry for developing his version of olefin metathesis catalysts. Although R3Ta=CHC(CH3)3 has been vigorously studied and appears in textbooks, its exact mechanism of formation has been a mystery. Researchers including Schrock have guessed that TaR5 forms as an intermediate between the starting material TaR3Cl2 and R3Ta=CHC(CH3)3. Zi-Ling (Ben) Xue of the University of Tennessee, Knoxville, reported fleeting glimpses of TaR5 in experiments 14 years ago, but there hasn't been unambiguous evidence to support the sitings. Xue, Julia K. C. Abbott, and Liting Li now provide definitive proof from their studies on deuterated versions of the compounds in the reaction sequence, which are more stable and easier to characterize (J. Am. Chem. Soc. 2009, 131, 8246). One of Xue and coworkers' key observations confirms that R3Ta=CHC(CH3)3 indeed forms via abstraction of an α-hydrogen atom from one neopentyl group that is carried away by a second neopentyl group. "It is nice to see a problem that has been out there for 35 years finally solved," Schrock says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter