Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Nitroxyl Lights Up Copper Complex
Copper-based fluorescent probe is a selective indicator for HNO in biological systems
by Celia Henry Arnaud
April 12, 2010
| A version of this story appeared in
Volume 88, Issue 15
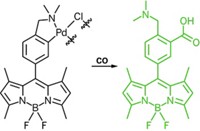
Nitric oxide might be the best known reactive nitrogen species relevant to biology, but it’s not the only one. Nitroxyl (HNO) is the reduced, protonated analog of NO and has several proposed physiological roles. But until now there hasn’t been a good way to detect HNO in biological systems. Stephen J. Lippard and Joel Rosenthal of Massachusetts Institute of Technology are reporting a copper-based fluorescent dye system that is selective for HNO (J. Am. Chem. Soc., DOI: 10.1021/ja909148v). The probe consists of a boron dipyrromethene (BODIPY) fluorescent dye connected to a copper(II) metal center. The copper’s ligand is made up of a tertiary nitrogen with two 2-pyridylmethyl substituents and a triazole substituent; the triazole serves as a bridge to the BODIPY group. This design minimizes the distance between the fluorescent reporter and the copper, ensuring the metal quenches the fluorescence in the system’s “off” state. When HNO is present, it reduces the copper, and the BODIPY group lights up. The researchers exposed the new complex to the nitroxyl anion donor Angeli’s salt (Na2N2O3), which generates HNO, and observed a rapid fourfold increase in fluorescence emission. The probe also responds to HNO in cell culture.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter