Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
NO2- Created
The periodic table has once again yielded a new form of a diatomic molecule
by Stephen K. Ritter
July 5, 2010
| A version of this story appeared in
Volume 88, Issue 27
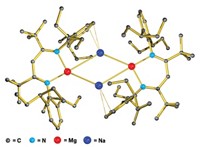
The periodic table has once again yielded a new form of a diatomic molecule—the radical dianion of nitrogen oxide, NO2– (Nat. Chem., DOI: 10.1038/nchem.701). Nitric oxide, NO, is a hybrid of N2 and O2 and exhibits extensive biochemical, industrial, and atmospheric chemistry. The unpaired electron on NO makes it highly reactive and easy to oxidize to NO+ and reduce to NO– , which are electronically analogous to N2 and O2, respectively. A year ago, William J. Evans of the University of California, Irvine, and colleagues isolated the reactive triply reduced N2 3– radical between two yttrium atoms in a diyttrium complex, expanding the collection of N2 species (C&EN, July 27, 2009, page 13). N2 3– is electronically analogous to the superoxide radical O2 – , an ion that plays important roles in immune system biochemistry. The discovery prompted Evans and his colleagues to try to make NO2– , the hybrid-species analog. The researchers knew the diyttrium N2 3– complex is a very strong reducing agent, which gave them the idea to react it with NO. When they did, the NO2– complex shown formed immediately. “After all the decades of research on NO, it is quite surprising to find a new oxidation level for this simple diatomic molecule,” Evans says.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter