Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Metal-Methane Insertion Milestone
Microwave spectroscopists have captured the first detailed structure of a transition metal inserted into a C–H bond of methane
by Stephen K. Ritter
December 6, 2010
| A version of this story appeared in
Volume 88, Issue 49
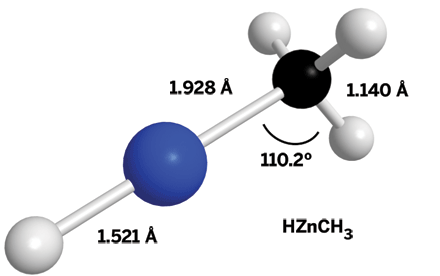
Researchers at the University of Arizona led by Lucy M. Ziurys have captured the first detailed structure of a molecule formed by insertion of a transition metal into a C–H bond of methane (J. Am. Chem. Soc., DOI: 10.1021/ja106121v). The achievement gives new insight into the mechanisms of catalytic C–C and C–H bond activations that are important in organic synthesis. Scientists have previously tried multiple approaches for obtaining a structural snapshot of metal-methane insertion complexes. Those efforts have been limited until now to photochemical reactions in low-temperature inert-gas matrices or gas-phase molecular beam experiments, which can help researchers tell that metal insertion has taken place but can’t provide exact structural details about the insertion products. Ziurys and coworkers produced HZnCH3 in the gas phase by reacting Zn(CH3)2 with H2 and CH4 in an electric discharge or by reacting zinc vapor with CH4 in an electric discharge. The researchers determined HZnCH3’s precise structure by examining zinc, carbon, and hydrogen isotopic variations in high-resolution rotational spectra recorded using a combination of microwave spectroscopy techniques. They found that HZnCH3 is relatively stable, has covalent H–Zn and Zn–C bonds, and likely forms by direct Zn insertion into a C–H bond rather than first forming ZnH or ZnCH3 species.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter