Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Prenylation With A Twist
Biochemistry: Enzyme catalyzes attachment of hydrophobic modifier to peptides in a new way
by Carmen Drahl
August 8, 2011
| A version of this story appeared in
Volume 89, Issue 32

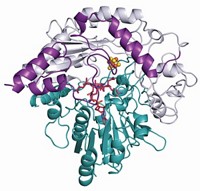
A newly discovered enzyme that fastens prenyl groups to peptides works in an unusual way, according to a research report (J. Am. Chem. Soc., DOI: 10.1021/ja205458h). The enzyme could complement the tool kit for constructing molecules with therapeutic potential through genetic engineering.
Linking prenyl groups, which are hydrophobic moieties derived from the five-carbon compound isoprene, to a protein or a small molecule is thought to facilitate attachment to cell membranes and is an important way of modifying biological activities, such as signal transduction or quorum sensing. But chemists have been unclear as to how prenylation occurs on a subset of natural products—peptides made by ribosomes. Now, graduate student John A. McIntosh and Eric W. Schmidt of the University of Utah; Satish K. Nair of the University of Illinois, Urbana-Champaign (UIUC); and colleagues have found an enzyme from a new family that carries out prenylations on cyclic ribosomal peptide natural products.
They also find that the enzyme, which they called LynF because it comes from the marine cyanobacterium Lyngbya aestuarii, acts by a novel mechanism. Normally, prenyl-fastening enzymes attach the prenyl group to a specific atom where it stays put. But LynF instead attaches the prenyl group first to an oxygen atom of tyrosine residues, and the resulting adduct then undergoes a spontaneous Claisen rearrangement to form a product prenylated at a tyrosine carbon atom. Such a Claisen-containing route to natural products was first proposed in the 1970s, but experimental evidence for the proposal was lacking until now, Schmidt says.
If the LynF enzyme proves promiscuous in its choice of substrates, it could be possible to genetically manipulate the enzyme and its natural product synthesis pathway for making potential drug leads in bacteria such as Escherichia coli, Nair says. “There’s a huge class of ribosomally synthesized cyclic peptides with antibiotic activity,” including the thiopeptides and the lantibiotics, Nair says, and this would be one potential area of application. The Schmidt group is now checking the extent of LynF’s promiscuity along with that of other enzymes in its family.
Biochemist Wilfred A. van der Donk of UIUC, who was not involved with the research, says the most interesting part about this work is that the LynF enzyme’s amino acid sequence is completely different from that of other prenyl-transferring enzymes.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter