Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Ocean Acidification’s Decalcifying Effects Explained In Detail
by Stephen K. Ritter
August 22, 2011
| A version of this story appeared in
Volume 89, Issue 34
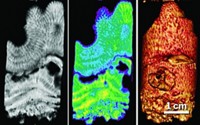
The negative response of corals and mollusks to ocean acidification stemming from increasing atmospheric carbon dioxide levels and warming temperatures varies widely, which has perplexed scientists because they have not been able to pin down general patterns for how the shells and skeletons of calcifying sea creatures grow and dissolve under lower pH conditions. Studies led by Riccardo Rodolfo-Metalpa of the University of Plymouth, in England, now provide clearer results by showing that the animals’ tissues and organic skinlike layers play a major role in protecting their calcium carbonate structures (Nat. Clim. Change, DOI: 10.1038/nclimate1200). The researchers transplanted corals, limpets, and mussels in the ocean off the coast of Italy near Mount Vesuvius, where CO2 bubbles up from the seabed as a result of volcanic activity. The test area serves as a natural laboratory to see how the animals respond to acidifying conditions. They also studied animals exposed to 45Ca-labeled water in the lab, which allowed them to measure CaCO3 formation and dissolution rates under varying conditions. The team observed that the creatures generally calcify more quickly at higher CO2 levels, but their unprotected surfaces dissolve away faster at lower pH, especially in combination with warmer water temperature. The net detrimental decline in calcification depends on how much protection individual species have.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter