Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Antagonistic Catalyst Turns Tandem Reaction
Controlled installation of acidic and basic functional groups on silica produces a two-in-one catalyst
by Stephen K. Ritter
September 19, 2011
| A version of this story appeared in
Volume 89, Issue 38
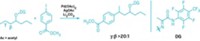
Chemists have succeeded in mimicking antagonistic acid and base catalytic functions observed in some enzymes by synthesizing a dual-purpose heterogeneous catalyst capable of carrying out one-pot tandem reactions (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201101449). Soluble homogeneous catalysts that combine chemically hostile functional groups have been made successfully, but synthesizing similar bifunctional solid catalysts “is no easy task,” saysGadi Rothenberg of the University of Amsterdam, who led the research team with colleague N. Raveendran Shiju. The trick, Rothenberg says, is to install the acid and base groups far enough apart so they don’t neutralize each other. To accomplish that, the researchers first grafted propylamine groups, –(CH2)3NH2, onto mesoporous silica and then immobilized phosphotungstate groups, –(CH2)3NH3[H2PW12O40], on half of the amines. The team tested the catalyst by converting dimethoxymethylbenzene to trans-1-nitro-2-phenylethylene. The tandem reaction begins with an acid-catalyzed deacetalization to form benzaldehyde, followed by a base-catalyzed Henry reaction with nitromethane to give the nitro product. The catalyst could be used for multiple types of reactions, Rothenberg adds, noting that it can be made primarily acidic, primarily basic, or equally acidic and basic by controlling the acid:base active-site ratio.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter