Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Reimagining life’s chemical engines
Biochemistry: Damaged DNA building block hints at the origins of life’s redox catalysts
by Carmen Drahl
September 19, 2011
| A version of this story appeared in
Volume 89, Issue 38

By studying damaged DNA, researchers have gained new insight about the possible predecessors of protein-bound cofactors, which help carry out the essential chemical reactions of life. They propose that in the so-called RNA world—an early period of life on Earth based on RNA instead of DNA—basic biochemistry might have been catalyzed by modified RNA building blocks, such as guanine.
The hypothesis opens up a possible different dimension to the RNA world. “We’re asking the question—what were the plausible first steps” on the road to the small-molecule helpers of protein biochemistry today? says lead researcher Cynthia J. Burrows of the University of Utah.
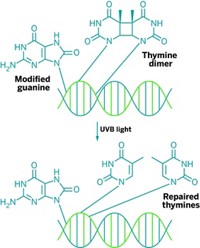
Burrows and graduate student Khiem Van Nguyen have observed that 8-oxo-7,8-dihydroguanine, an oxidatively damaged version of the base guanine, can mimic a flavin, a type of cofactor (J. Am. Chem. Soc., DOI: 10.1021/ja2072252). Specifically, the damaged base can catalyze the repair of another type of DNA damage—light-mediated dimerization—something a flavin-containing enzyme can also do.
The researchers think the bases found in DNA and RNA were a likely evolutionary starting point for cofactors such as flavin adenine dinucleotide, a key player in the production of the energy source adenosine triphosphate. But plausible first steps on that evolutionary journey haven’t been defined.
Flavins are effective catalysts in part because of their low redox potentials. Burrows, an expert in DNA damage, knew that 8-oxo-7,8-dihydroguanine possessed a redox potential closer to a flavin’s than to those of canonical bases. So she and Nguyen incorporated the damaged base into synthetic DNA or RNA strands that contained a nearby thymine dimer, another type of DNA damage. With exposure to ultraviolet light, the modified guanine catalyzed cleavage of the thymine dimer, thereby repairing the damage.
“I’d gotten used to the idea that any changes in the structure of DNA that could cause mutations are ‘damage’ and are bad, but that’s an outlook based on life as we know it today,” Burrows says. “I’m starting to think that chemical modifications of bases could have been very useful 4 billion years ago,” before proteins emerged, she says.
Burrows says the next steps will be to try to evolve the possible proto-flavin further in the lab, examine reaction conditions for the repair process that are more plausible for a primordial Earth, and explore reactivity of other modified bases.
“If RNA catalysis predated protein catalysis, then what predated the enzyme cofactors that endow these [protein] biopolymers with their breadth of function?” asks Steven E. Rokita, who studies nucleic acid reactivity at the University of Maryland, College Park. Burrows and Nguyen’s work “presents a fascinating challenge to those interested in prebiotic chemistry and the RNA world,” he says. Moreover, the study also finds a beneficial function for a common type of DNA damage, he adds.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter