Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Personalizing Nanomedicine
Researchers strive to tailor patient care by developing nanoparticles called theranostics that can simultaneously monitor and treat disease
by Lauren K. Wolf
September 26, 2011
| A version of this story appeared in
Volume 89, Issue 39
Thanks to the sci-fi movies and novels prevalent in popular culture, the term “futuristic medicine” might conjure images of intelligent robots performing complex surgeries and miniaturized ships navigating the body to seek and destroy lurking malignancies.

To some, Jelena M. Janjic’s hope for the future of medicine might also seem fanciful: She envisions that a patient will someday be treated with tiny targeted particles loaded with drugs, and that after administering the particles, doctors will be able to look inside the patient to see if the drugs have hit their mark—whether that target is an organ, a tumor, or a joint. From there, the physicians can adjust the dosage or type of these nanoparticles as needed.
More specifically, Janjic, a professor of pharmaceutics at Duquesne University, sees a time when a breast cancer patient might go into a clinic and receive a transdermal patch loaded with therapeutic nanoparticles. “You apply this to the skin, and the nanoparticles penetrate,” she says. Then, assuming the guise of a physician, she adds, “I tell you to come back in three days for imaging to see how well the patch is providing the particles to your tissue.”
If the breast tumor is close to the surface of the skin, doctors could use near-infrared imaging, a tissue-penetrating optical technique, to monitor the particles. For a deeper malignancy, magnetic resonance imaging (MRI) might be the method of choice. To be detected by these methods, the particles would need either to have an intrinsic signal or be tagged with a signaling compound.
After the imaging, Janjic says, doctors could assess how much of the drug delivery vehicle reached the tumor at a given concentration. “From that point, I can say we need to dose once per month, or if it isn’t the level I want, I can increase the dose or frequency while monitoring the progress of treatment,” she says.
This futuristic scenario for personalized medicine isn’t that far-fetched. Combining therapeutics and diagnostics into a single platform is a concept that has been growing in popularity in the past decade, particularly as the U.S. Food & Drug Administration has started approving some nanomaterials as either drugs or imaging agents.
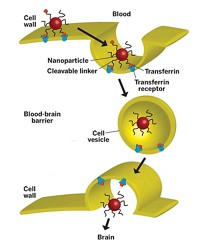
Nanoparticles are ideal for all-in-one therapy and diagnostic packages because their small size allows them to slip into living cells, and their large surface-to-volume ratio enables them to hold a relatively high concentration of drugs and imaging agents. So far, though, nanoparticles intended to have both capabilities—called “theranostics”—haven’t made it to clinical trials. Before these materials can become a reality, researchers such as Janjic, who are hard at work devising strategies to combine therapeutics and diagnostics into a single nanosized platform, have a number of obstacles to overcome.
Coined in 2002, “theranostics” describes a research field that is “hot” right now, says Theresa M. Reineke, a chemist at the University of Minnesota and coauthor of a recent Bioconjugate Chemistry review on the topic (DOI: 10.1021/bc200151q). As originally defined, a theranostic is any material that can both diagnose and treat a disease and that can possibly monitor the efficacy of therapy on a personalized level.
“But now researchers are being more creative and combining therapeutics plus some type of diagnostic capability in general,” Reineke explains. The drug and the diagnostic method can be applied separately. Under this broader definition, for example, Genentech’s monoclonal antibody for breast cancer treatment, Herceptin, is a theranostic when combined with HercepTest, an antigen-based staining assay from diagnostics firm Dako. Doctors run the HercepTest on biopsied tumor samples to determine whether patients will respond to treatment.
How theranostics will be defined in the future remains to be seen, but many scientists are sticking with the original, narrower definition, investing their time and effort in developing single platforms—such as gold and iron oxide nanoparticles, polymer beacons, liposomes, and nanoemulsions—for therapy and diagnosis.
These researchers have their work cut out for them, according to Sanjiv S. (Sam) Gambhir, director of the molecular imaging program at Stanford University’s School of Medicine, because getting nanoparticle-based theranostics into humans will easily take a decade. Gambhir, who has tested and developed a lot of imaging agents in humans over the years, says regulatory issues will slow down nanoparticle-based theranostics’ advance to the doctor’s office. “FDA is still getting comfortable with how it should set standards for nanoparticles in humans,” he says.
Small-molecule synthesis and purification yields compounds that are uniform from batch to batch, he points out. Nanomaterial synthesis, on the other hand, could produce a range of particle or vesicle sizes and shapes, he says. The fabrication process might also generate materials with a slightly different number of drugs, targeting molecules, or imaging agents on their surfaces. “These are newer types of issues that FDA is not sure how to treat,” Gambhir says.
For this reason, Gambhir’s group has been taking small, measured steps toward the goal of theranostics. “We’ve been first optimizing the delivery and diagnostic part of the equation” before adding the therapeutic component, he says. The researchers have started by testing the toxicity of gold nanoparticles coated with a layer of Raman-active molecules, followed by a layer of polyethylene glycol (PEG)-capped silica. After injection into mice, the particles caused slight inflammation in the liver for two weeks but had no toxic effects, eventually clearing from the rodents’ systems (Sci. Transl. Med., DOI: 10.1126/scitranslmed.3001963).
The eventual plan for these particles is to link a molecule to the PEG moieties that targets epidermal growth factor receptor (EGFR) and other protein targets overexpressed in cancerous cells. For colon cancer patients, doctors could use an endoscope to deliver light into the body and, via surface-enhanced Raman imaging, see whether the particles find their mark in a tumor.
Gambhir says he also wants to use this type of particle to treat and image gliomas, tumors that thread tiny fingerlike projections into brain tissue and are difficult to remove during surgery. With these theranostics, however, doctors could inject the targeted nanoparticles into a patient before surgery and then shine laser light on the tumors, Gambhir says. The light would excite a surface plasmon resonance, or collective oscillation of electrons, in the gold, heating and killing the cancerous cells—a process called photothermal therapy.
Younan Xia has also devised a gold-based theranostic for treating disease. The professor of biomedical engineering at Washington University in St. Louis, says that, like a lot of other scientific discoveries, his gold nanocages started out as a curious material without an immediate application. Ten years later, the hollowed-out, porous particles are finding use as both imaging agents and drug delivery vehicles.
When presenting his work at last month’s American Chemical Society national meeting in Denver, Xia showed that the nanocages can be loaded with chemotherapeutics such as doxorubicin and coated with a heat-sensitive polymer, poly(N-isopropylacrylamide). At room temperature, the “smart” polymer chains are extended and tangled, plugging up the slats in the nanocage and holding the cancer drug inside. When irradiated with a near-infrared laser, the chains collapse from the photothermal heat gold generates, and doxorubicin is released (Nat. Mater., DOI: 10.1038/nmat2564).
The great thing about using gold-based theranostics, Xia says, is that they have inherent imaging and photothermal therapy capabilities. Liposomes, polymer vesicles, or other organic-based systems have to be labeled with radioactive isotopes or heavy-metal tags to be used as contrast agents. Gold, though, is a natural photoacoustic agent when irradiated with light, generating an acoustic wave that shows up in ultrasound images. It can also be observed in computed tomography (CT) scans because of its ability to strongly attenuate X-rays.

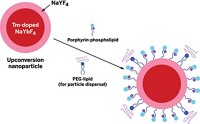
But gold is expensive. On the basis of cost concerns, iron oxide nanoparticles are a promising alternative theranostic platform. Like gold nanoparticles, these tiny materials have inherent imaging and therapy capabilities: They are superparamagnetic, making them detectable by MRI, and they can be heated with an alternating magnetic field to destroy tumors—a therapy known as hyperthermia.
Xiaoyuan (Shawn) Chen, editor-in-chief of the online journalTheranostics, which was launched early this year, has worked extensively with iron oxide nanoparticles. In addition to loading the tiny materials with chemotherapeutics, he and his team at the National Institute of Biomedical Imaging & Bioengineering upgraded the materials from having a single imaging mode—MRI—to having multiple modes. By coupling the particles to the near-infrared fluorescence dye Cy5.5 and a compound chelated to the radioisotope 64Cu, the researchers enabled the tiny materials’ detection in the tumors of mice via fluorescence imaging and positron emission tomography (PET), respectively (Biomaterials, DOI: 10.1016/j.biomaterials.2009.08.045; J. Nucl. Med., DOI: 10.2967/jnumed.108.051243).
Imparting multiple imaging modalities to different parts of these nanomaterial-based platforms could help address what Chen thinks is a major challenge for theranostics. “These particles are composites,” Chen explains. “They have several components,” such as drug molecules and polymer coatings. For iron oxide, MRI reflects only the position of the particle core. “But you also have the coating and the drugs—what’s happening to those?” he asks. For these nanomaterials to be used successfully in personalized medicine, he adds, “you need to have other imaging methods to follow the release and efficacy of the drug.”
Not only does Chen think iron oxide nanoparticles are promising theranostic platforms, but he also likes the odds on synthetic polymer nanomaterials. “Particles based on these have a good opportunity to move into the clinic,” he says. They can be produced reliably and at low cost.
The University of Minnesota’s Reineke thinks so too. Her team has designed nucleic acid delivery theranostics that are created by complexing polycation imaging beacons with DNA. The beacons contain repeating units of oligoethyleneamines and chelated gadolinium, a heavy-metal MRI contrast agent. When the polymers form a complex with the DNA, the beacons coil up into colloidal nanoparticles that compact the double-helical macromolecule and neutralize its negative charge, thereby enabling it to be taken up by cells. Reineke’s team demonstrated that these so-called polyplexes are capable of delivering fluorophore-labeled plasmid DNA into cancerous HeLa cells in vitro (Proc. Natl. Acad. USA, DOI: 10.1073/pnas.0904860106).
In the case of nucleic acid therapy, Reineke says, her team is working on delivering small interfering RNAs (siRNAs) with the polymer beacons to disrupt transcription of genes involved in tumor formation. Although this is the next step for her team, Reineke says, they’ll first need to know more about the delivery mechanism to optimize the theranostic. “From the fluorescence microscopy images, it appears that the polymer beacon is dissociating from the nucleic acid in the cells’ cytoplasm, but more investigation is needed to find out how and where this is occurring,” she adds.

Aside from understanding the mechanism and distribution of drug release from theranostics, researchers are also at some point going to have to tackle the challenge of optimizing dosage, Reineke says. The amount of drug needed to treat a patient doesn’t always match the concentration of imaging agent required to monitor a theranostic’s accumulation in tissue. For instance, doctors administer cancer drugs such as doxorubicin at a dose in the range of milligrams per kilogram of body weight for optimal treatment results, whereas imaging agents such as radioactive isotopes can be used only at levels less than micrograms per kilogram because of toxicity issues.
At the ACS national meeting, Ki-Bum Lee described his group’s efforts to increase theranostics’ imaging signal and decrease the amount of therapeutic required to effectively treat patients. Lee and his team at Rutgers University have fabricated iron cobalt particles coated in graphite and dextran to both deliver siRNA to tumor cells and kill them via hyperthermia treatment. The researchers chose iron cobalt for its enhanced superparamagnetism—and thus MRI contrast ability—over iron oxide, Lee says. They demonstrated in vitro that their particles, when conjugated to an EGFR-targeting antibody as well as to an EGFR-gene-targeting siRNA, significantly increased death among brain tumor cells when combined with hyperthermia treatment (Small, DOI: 10.1002/smll.201100012).
Advertisement
Ultimately, though, whether nanomaterial-based theranostics become a reality could depend on the expense and complexity of their development. These parameters affect what companies in this field will push to the clinic and what insurance companies will pay for a treatment regimen, Reineke says. Incorporating multiple imaging techniques such as MRI and PET is expensive, she adds.
Duquesne’s Janjic, who has the background of a pharmacist and medicinal chemist, thinks that over the next few years some of the complex theranostic strategies being published rampantly in chemistry journals will fall out of contention in the race toward personalized medicine. “For me, something that’s too difficult to make or too complex to sustain in large-scale production is not what we are interested in,” she says. “We want something that is simple, elegant, and still does the job.”
At the ACS meeting, Janjic presented her team’s work on developing fluorocarbon-based theranostics. These nanoemulsions have a 19F-labeled fluorocarbon core for MRI imaging surrounded by a hydrocarbon phase containing a near-infrared imaging agent and a drug. The emulsion is stabilized with an outer layer of surfactant.

The therapeutic Janjic is currently incorporating into her formulation is celecoxib, a nonsteroidal anti-inflammatory drug that targets the enzyme COX2. “We are trying to suppress COX2,” which is overexpressed in breast cancer patients, “and suppress it long enough that tissue inflammation caused by solid tumors is suppressed,” she says. “We hope that will help tumor regression” when combined with other therapies, she adds.
Janjic, whose motivation to find more effective breast cancer therapies stems from the loss of two family members to the disease, has high hopes for her theranostic strategy as well as others in development. What she learned from her loved ones’ ordeals is that “not every therapy works for everyone,” she says. “That’s the promise of theranostics,” she adds. “You can tailor the therapy to the stage and type of disease, and you can fine-tune the treatments for the best outcome.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter