Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Carbon Monoxide Boosts Gold Catalysis
Classic catalyst poison unexpectedly found to enhance electrocatalytic reaction
by Mitch Jacoby
December 12, 2011
| A version of this story appeared in
Volume 89, Issue 50
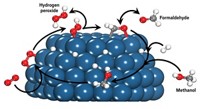
Carbon monoxide adsorbed on gold enhances the precious metal’s ability to catalyze oxidation of alcohols in an electrochemical cell, according to researchers at Leiden University, in the Netherlands (Nat. Chem., DOI: 10.1038/nchem.1221). CO is a classic example of a poison to many transition-metal catalysts because it typically binds strongly to metal active sites, denying access to reactant molecules. Not only does the study add to the growing list of catalytic transformations mediated by gold, long considered an inert material, but it also suggests “an unexpected and counterintuitive role for CO in electro-oxidation reactions, namely that of catalyst promoter,” says Marc T. M. Koper, who led the study. Paramaconi Rodriguez, Youngkook Kwon, and Koper found that for a gold electrode immersed in an aqueous alkaline solution, the minimum applied potential required to oxidize methanol (mainly to formaldehyde and formic acid) is significantly lower when CO is adsorbed on the gold than when it is absent. The team studied reactions of various alcohols and proposes that CO’s presence promotes binding of OH species, which catalyze a key hydrogen elimination step in alcohol oxidation.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter