Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Molecular Machine’s Workings Visualized
Chemical labeling and microscopy technique demonstrates its power on the spliceosome
by Stu Borman
December 19, 2011
| A version of this story appeared in
Volume 89, Issue 51

COVER STORY
Molecular Machine’s Workings Visualized
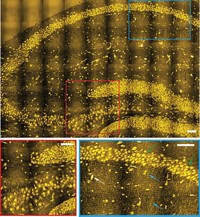
A fluorescence microscopy method that marks biomolecules with organic dyes made it possible this year to understand the workings of complex biomolecular processes at the single-molecule level. The technique was developed by Melissa J. Moore of the University of Massachusetts Medical School, Jeff Gelles of Brandeis University, Virginia W. Cornish of Columbia University, and coworkers (C&EN, March 21, page 48; Science, DOI: 10.1126/science.1198830). The research team demonstrated the approach’s capabilities by monitoring the assembly of the spliceosome, a huge protein-RNA complex that edits newly transcribed mRNA to create mature mRNA, which encodes the chemical blueprints for proteins. Postdoc Aaron A. Hoskins in Moore’s lab tagged yeast spliceosome components with organic fluorophores using a technology developed by Cornish’s group. The photon output of the different-wavelength fluorescent tags was 10 times as great as that of conventional fluorescent proteins, enabling greater sensitivity for simultaneous detection of the individual spliceosome components. Hoskins then used multilaser colocalization single-molecule spectroscopy developed by Gelles’ group to monitor the assembly of spliceosomes. As flashes of different colors of light were given off, he was able to track each component in real time as it joined the assembly. The researchers believe the method will be useful for tracking other complex bioprocesses, such as DNA replication and protein translation.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter