Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Bacteria Bearing Messages
Chemical Communications: Method permits encoding, decoding text messages with the use of living organisms
by Stu Borman
October 7, 2011

Aiming to enhance the interface between chemistry, biology, and information technology, a research team has developed a technique to encode messages in patterns of bacteria and reveal them through fluorescence.
“We think our technique can potentially be used for easy-to-read biological barcoding, as a deterrent to counterfeiting, or, of course, for secret communications,” says postdoc Manuel A. Palacios of Tufts University, first author of a paper describing the approach (Proc. Natl. Acad. Sci. USA, DOI: 10.1073/pnas.1109554108). The study was carried out by Palacios, Tufts chemistry professor David R. Walt, Harvard University chemistry professor George M. Whitesides, and coworkers.
The technique’s advantage is the use of a visible, easy-to-read output for the encoded information—the output being a pattern of fluorescent signals produced when the bacterial messengers are grown in an array. Nonbiological information has been encoded in the DNA of microorganisms before, mostly for barcoding purposes, but expensive sequencing or other molecular biology laboratory techniques were needed to read the messages. With the new approach, all that is needed to read an encoded message is a light diode source to excite fluorescence and a camera to record and interpret it.
The technique was conceived as part of the Defense Advanced Research Projects Agency’s (DARPA’s) Chemical Communications program, which supports the development of “self-powered chemical systems that can encode an input string of alphanumeric characters (a message), convert the message to a modulated optical signal, and transmit it repetitively to a receiver,” according to the agency.
Earlier, Whitesides, Walt, and coworkers developed “infofuses,” which act as infrared signal flares to send messages to remote viewers (Proc. Natl. Acad. Sci. USA, DOI: 10.1073/pnas.0902476106, and C&EN, June 1, 2009, page 8). Now, the group has extended that concept by developing a signaling mechanism mediated by bacteria instead of being propagated directly through space.
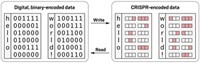
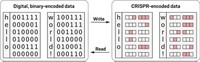
The technique makes use of seven strains of bacteria that have been genetically engineered to express different fluorescent proteins. By assigning a color pair to a letter, number, or other character, the seven strains can encode 49 alphanumeric characters.
To compose a message, the sender spots on a membrane the pattern of bacterial pairs that spells the information. At the decoding stage, the recipient stamps the message-containing membrane into a growth medium, induces fluorescence expression, and reads the pattern of fluorescent dots visually according to the agreed-upon color code. Walt and coworkers call this way of transmitting information “InfoBiology,” and they call the technique “SPAM” (Steganography by Printed Arrays of Microbes). “Steganography” is hidden messaging.
One could argue that there is no preexisting problem that the SPAM technique solves, because there are already many nonchemical ways to transmit secret or encoded information. In response, Walt explains that SPAM meets DARPA’s requirements for a nonelectronic encryption system and has advantages over current encoding systems. Notable among these is its many layers of security, such as the code, the growth medium, the timing needed to express the signals (which can be made variable), and the ability to encode multiple messages in the same array (by using bacteria resistant to different antibiotics, for example). Applications of SPAM, such as barcoding and authentication, would benefit not only from the messages’ invisibility but also from the near impossibility of deciphering them given the multiple layers of security, he says.
Computational biologist Masanori Arita of the University of Tokyo, who has worked on similar projects, says SPAM’s current 49-character repertoire is somewhat limiting. Nevertheless, he says, the technique is “an elegant way to visualize human-encoded information and brings the world of biotechnology and computation much closer.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter