Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Education
Microscale Chemistry
January 9, 2012
| A version of this story appeared in
Volume 90, Issue 2
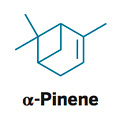
Correction:
Dec. 5, 2011, page 35: The dehydro form of pinene was shown; one of the double-bonded isomers, α-pinene, is shown here.
I read Maureen Rouhi’s “Minimalist Chemistry” article with interest (C&EN, Oct. 3, 2011, page 41). The more I read the article, however, the more I thought, “What is so new with the procedures being described?” The techniques of microscale chemistry have been being used for years in the U.S.
I personally have presented three papers at ACS national meetings on this subject. At North Central College in the early 1980s we were using plastic well plates with 12 wells in our introductory courses. There are many advantages to using the well plates for electrochemistry, acid-base, qualitative screening, and many other tests. Some instructors in our DuPage County High Schools are even using the 96-well plates.
In the early 1980s, the National Microscale Center at Merrimack College began presenting National Science Foundation workshops in organic, general, and inorganic chemistry. These one-week workshops were well received and helped many educators incorporate the techniques in their classes. A literature search of the use of small-scale chemistry reveals that instructors were beginning to use the smaller scales in their classes and also cut quantities in the early 1930s. John Yoe of the University of Virginia published several papers in the 1930s. I have even attended ACS national meeting sessions where the presenters brought adequate supplies for the attendees to participate in the small-scale reaction demonstrations.
I am happy that Japanese educators are being creative and continuing to teach under stressful situations, but to publish their techniques as new or unique is not quite true. I feel that the author should have mentioned some of the earlier work done in the U.S. for a number of years.
By Anne T. Sherren
Lombard, IL





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter