Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Getting Enzymes To Make Cyclopropanes
Researchers engineer cytochrome p450 enzymes to do olefin cyclopropanation
by Sarah Everts
December 24, 2012
| A version of this story appeared in
Volume 90, Issue 52
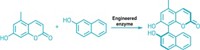
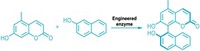
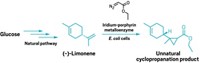
Researchers have trained an enzyme to perform a reaction not seen in nature but could be used for making pharmaceuticals, insecticides, and a portfolio of hormones and pheromones. California Institute of Technology’s Frances H. Arnold and her colleagues engineered a bacterial enzyme called cytochrome P450BM3 to turn olefins into cyclopropanes. That reaction previously could be carried out only with inorganic catalysts (Science, DOI: 10.1126/science.1231434). Cytochrome P450BM3 is a member of a protein family famous for catalyzing hydroxylations and other oxidative transformations. The team reasoned that it could be adapted for olefin cyclopropanation. Using a library of mutant P450BM3 enzymes, they found a handful that could perform the desired reaction, but with low yield and poor enantiospecificity. When the team tweaked amino acids in several P450BM3 enzymes’ active sites , they observed reactions with high turnover, enantioselectivity, and diastereoselectivity. “The established reaction promiscuity of natural enzymes and the surprising ease with which cyclopropanation activity could be installed into P450BM3 suggest that this approach will be useful for other synthetically important transformations for which biological counterparts do not yet exist,” the authors note.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter