Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
A Drug Carrier That Dissolves In Cells
Cellular Delivery: Protein-based particles release nucleic acids by dissolving inside cells
by Erika Gebel
May 15, 2012

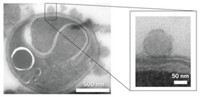
To develop vehicles to deliver drugs inside the body, researchers have tested a range of particles made from materials such as polymers and proteins. Proteins seem ideal because they’re inherently biocompatible. But they’re also soluble in water, so the proteins dissociate from the particles in blood. Now researchers have tweaked protein particles so they hold together until they reach the inside of a cell, where they then dissolve and release their cargo (J. Am. Chem. Soc., DOI: 10.1021/ja302363r).

Nucleic-acid-based therapeutics, such as small interfering RNAs, are highly charged, making it difficult to encapsulate them inside a particle for delivery. Joseph DeSimone of the University of North Carolina, Chapel Hill, thought a polar material, such as a protein, would work best. “What’s a better polar matrix than a protein?” he asks. But to combat the protein particles’ solubility, DeSimone wanted to develop a way to make them insoluble outside of a cell, but soluble once inside.
His team achieved this solubility switch by stitching together the proteins once they had formed a particle. They used a compound called dithio-bis(ethyl 1H-imidazole-1-carboxylate) (DIC), which crosslinks proteins via their amine groups. This crosslinking holds the proteins together and prevents the particle from dissolving. But the crosslinking compound contains a disulfide bond that breaks in reducing environments, such as inside a cell, explains DeSimone. So once the particle slips into a cell, it breaks apart, dissolves, and releases its cargo. In addition, when the disulfide bonds break, the remaining halves of the DIC molecule fall off the amine groups, restoring the proteins to their unmodified state.
To test the protein-stitching technique, the researchers first made protein particles using a method they had developed previously (J. Am. Chem. Soc., DOI: 10.1021/ja8014428). They combined bovine serum albumin, an inexpensive protein, with glycerol and α-D-lactose, which act as glues to hold the protein matrix together. As test cargo, they mixed in a large RNA molecule that codes for the enzyme chloramphenicol acetyltransferase. To form the particles, the researchers poured this mixture into a mold to solidify. They pulled the protein particles from their molds with sticky paper and then dissolved the adhesive to release the particles.
The team then bathed the free particles in a solution of DIC to stitch them together. They added a compound called a transfection agent to the particles’ surface to help them pass through cell membranes. They exposed monkey kidney cells to the final particles to see if the particles could deliver the RNA. After four hours, the researchers lysed the cells. An enzyme-linked immunosorbent assay showed that the cells produced the enzyme, demonstrating that the particles successfully delivered the RNA. The particles were as efficient as a transfection agent alone, a standard method to deliver nucleic acids into a cell.
“It’s just beautiful,” says Paul Wender at Stanford University about the new technique. “What’s nice about the strategy is that once the particle gets inside the cell, it sheds the coating and becomes the original protein.” He thinks this opens up the possibility of using the protein matrix itself as a medication.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter